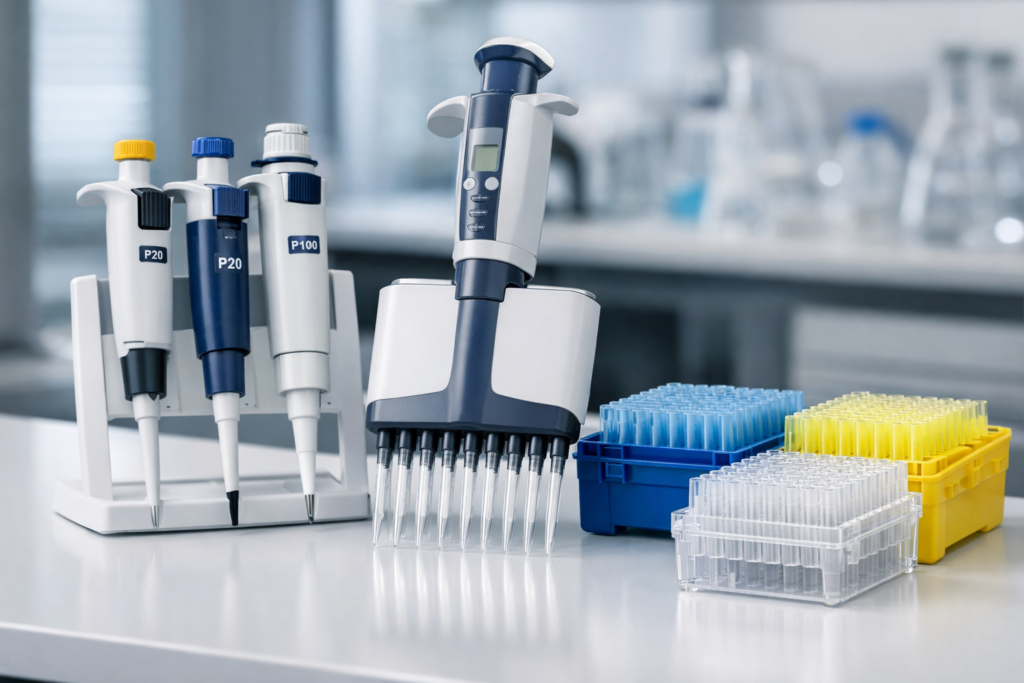
A pipette is a precision instrument designed to measure and transfer specific volumes of liquids in laboratory settings. Whether in clinical diagnostics, pharmaceutical research, biochemistry, or quality control environments, pipettes represent one of the most fundamental and frequently used tools in any modern laboratory. Despite their apparent simplicity-a tube with a graduated volume scale and a mechanism to draw and expel liquid-pipettes are sophisticated instruments whose accuracy, precision, and proper use directly impact experimental validity and scientific integrity.
The importance of pipettes in laboratory work cannot be overstated. In fields ranging from molecular biology to analytical chemistry, from clinical immunology to environmental testing, the reliability of pipetting directly determines whether results will be reproducible, statistically valid, and actionable. A seemingly minor error in liquid handling-delivering 99 microliters instead of 100-can invalidate PCR reactions, compromise assay results, or lead to incorrect clinical diagnoses. This is why pipettes are subject to rigorous international standards, regular calibration protocols, and extensive operator training in virtually every accredited laboratory worldwide.
The distinction between basic understanding and professional expertise in pipetting is substantial. While anyone can physically operate a pipette, mastering the technique requires knowledge of multiple factors: the specific type of pipette suited to the application, the properties of the liquid being transferred, environmental conditions affecting accuracy, proper ergonomic technique to prevent injury, and the principles underlying calibration and quality control. This comprehensive guide addresses all these dimensions, providing laboratory professionals, students, managers, and procurement specialists with the authoritative knowledge necessary to select, operate, and maintain laboratory pipettes effectively.
What Is a Pipette and How Is It Used in Laboratories?
A pipette is a volumetric measuring and liquid transfer instrument consisting of a calibrated tube with a narrow tip, a plunger mechanism (operated manually or electronically), and graduated volume markings or fixed volume settings. The fundamental principle underlying all pipettes is volumetric displacement: by moving a piston or plunger through a measured distance, a specific volume of air is displaced, creating a pressure differential that draws liquid into the pipette tip and subsequently releases it into a target container.
Pipettes serve several critical functions in laboratory workflows:
Quantitative Sample Preparation: In chemistry and biochemistry, pipettes enable the precise measurement and mixing of reagents and standards, essential for creating calibration curves, dilution series, and stock solutions. Analytical accuracy depends directly on the volumetric precision of these preparations.
Molecular Biology Applications: In molecular biology laboratories, pipetting is central to DNA and RNA work. PCR reactions, for example, typically involve combining template DNA, primers, polymerase, and dNTPs in precise ratios-a single pipetting error can result in complete reaction failure. Similarly, in DNA sequencing, library preparation, and cloning workflows, accurate liquid handling is non-negotiable.
Clinical and Diagnostic Testing: Hospital and clinical laboratories rely on pipettes for specimen processing, reagent dispensing, and quality control in hundreds of tests performed daily. In immunoassays, ELISA protocols, and automated clinical analyzers, pipetting accuracy directly affects diagnostic accuracy and patient care.
High-Throughput Screening: In drug discovery and genomics research, pipetting is often the rate-limiting step in laboratory throughput. Multichannel and electronic pipetting systems enable researchers to process thousands of samples in parallel, making large-scale studies economically feasible.
Pharmaceutical Development: In pharmaceutical quality assurance and manufacturing, pipettes are used for content uniformity testing, stability studies, and formulation development. Regulatory compliance (FDA, EMA, ICH guidelines) mandates that all pipetting be traceable to calibrated instruments.
Why accuracy matters in pipetting goes beyond the obvious: it connects directly to experimental power and reproducibility. When pipetting introduces excessive variability, researchers require larger sample sizes and more replicates to achieve statistical significance. This increases costs, consumes time, and may render studies unfeasible. Conversely, well-calibrated pipettes with proper technique reduce random error, enabling researchers to detect real biological effects with smaller sample sizes.
The difference between a trained technician and an untrained operator becomes apparent when comparing results. Studies consistently show that improper pipetting technique-incorrect angle, insufficient prewetting, excessive speed, or temperature mismanagement-introduces systematic errors that can bias results by 10-50% or more, depending on the liquid properties and pipette type.
Types of Pipettes Used in Laboratories
Laboratory pipettes are classified by several dimensions: the mechanism of operation (manual vs. electronic), the range and type of volumes measured (micropipettes vs. macropipettes, fixed vs. variable volume), the physical design (single-channel vs. multichannel), and the operating principle (air displacement vs. positive displacement). Understanding these distinctions is essential for selecting the appropriate instrument for any given application.
Manual Pipettes
Manual pipettes are operated entirely by hand, with the user controlling the piston movement through finger pressure on a button or plunger. This direct mechanical operation has been the foundation of laboratory liquid handling for over a century and remains widely used today.
Operating Principle: The user sets the desired volume using a rotating dial or digital readout, then positions the pipette tip into the source liquid. Pressing the plunger button to the first stop draws liquid into the tip through negative pressure; releasing the button allows atmospheric pressure to refill the pipette. To dispense, the plunger is pressed to the first stop again; pressing to a second stop (the “blow-out”) ejects any remaining liquid from the tip.
Advantages of Manual Pipettes:
- Cost-effectiveness: Manual pipettes are substantially less expensive than electronic models, often costing one-quarter to one-half the price. This makes them economically feasible for purchasing in larger quantities or for labs with budget constraints.
- Durability and Simplicity: With fewer electronic components, manual pipettes are more robust and require minimal maintenance. The mechanical design has proven reliable over decades, and repair is straightforward.
- Ease of Training: The mechanical simplicity of manual pipettes makes them ideal for training new laboratory personnel. The immediate feedback from hand operation helps users develop proper technique.
- No Power Requirements: Unlike electronic pipettes, manual pipettes require no batteries or electrical charging, eliminating downtime from power issues.
Limitations and Considerations:
- Operator-Dependent Variability: Manual pipetting introduces user-dependent variability. Different operators-or even the same operator on different days-may apply slightly different force and speed, resulting in volume variations.
- Repetitive Strain Risk: Repeated manual plunger operation thousands of times daily contributes significantly to repetitive strain injury (RSI), including carpal tunnel syndrome, tendonitis, and general hand pain.
- Slower Throughput: For applications requiring hundreds or thousands of pipetting steps (such as microplate assays), manual pipetting becomes prohibitively time-consuming.
- Accuracy Limits: While capable of meeting ISO 8655 accuracy standards, manual pipettes typically achieve repeatability coefficients of variation (CV) of 1-3%, whereas electronic pipettes routinely achieve <0.5% CV.
Manual pipettes remain appropriate for applications where speed and throughput are not priorities, budgets are constrained, or the particular workflow benefits from direct operator control.
Electronic Pipettes
Electronic pipettes incorporate motorized piston control, enabling programmable dispensing profiles and dramatically improved precision through consistent motor-driven motion. These instruments have become increasingly prevalent as costs have declined and their superior performance in high-throughput environments has become evident.
Operating Principle: Electronic pipettes use a stepper motor or other actuator to control piston movement with precision. The volume is typically set via digital buttons or touchscreen interface. Once the volume is set, pressing a single button triggers a preprogrammed sequence: the motor draws liquid into the tip at a controlled speed, holds it, and dispenses it at a controlled rate. Multiple channels can be controlled simultaneously in multichannel models.
Advantages of Electronic Pipettes:
- Superior Accuracy and Precision: Electronic pipettes maintain consistent piston speeds and pressures across repeated cycles, achieving coefficient of variation (CV) values typically <0.5%, compared to 1-3% for manual pipettes. This is particularly important in quantitative applications like qPCR.
- Reduced Ergonomic Strain: By eliminating repetitive manual plunger operation, electronic pipettes reduce the force and repetitive motion that cause RSI. Studies show that nearly 90% of researchers using manual pipettes more than one hour daily report hand or elbow pain, compared to substantially lower rates with electronic pipettes.
- Programmable Features: Electronic pipettes can be programmed with custom protocols, including prewetting steps (drawing and dispensing the liquid multiple times to equilibrate temperature), speed variation, and multi-step dispense sequences. These capabilities improve accuracy with challenging liquids and reduce errors.
- Higher Throughput: Electronic multichannel pipettes can dispense to 8, 12, or even 96 wells simultaneously, increasing throughput by 8-96-fold compared to single-channel manual operation.
- Reduced Operator Variability: Since the motor delivers consistent motion regardless of operator hand strength or experience, results are more reproducible across different users and shifts.
Disadvantages and Considerations:
- Higher Initial Cost: Electronic pipettes cost 2-5 times more than equivalent manual models, representing a significant capital investment.
- Battery/Charging Requirements: Electronic pipettes require regular charging or battery replacement. Incomplete charging or battery failure can interrupt workflows.
- Greater Complexity: More components mean more potential points of failure. Repair typically requires specialized technicians.
- Weight: Electronic pipettes are heavier than manual equivalents due to the motor and battery, which can contribute to fatigue during extended use (though generally less than the fatigue from manual plunger operation).
Electronic pipettes are increasingly considered standard equipment in research and clinical laboratories, particularly for applications demanding high accuracy, reproducibility, or throughput.
Micropipettes
Micropipettes are precision instruments designed to measure and transfer small liquid volumes, typically ranging from 0.5 microliters (μL) to 5000 microliters (5 mL). The term “micropipette” often refers specifically to variable-volume piston pipettes (as opposed to volumetric pipettes, discussed separately), though the distinction is somewhat arbitrary.
Volume Ranges and Types:
Micropipettes are categorized by their nominal (maximum) volume capacity:
- P1-P10: 0.5-10 μL (ultra-precision, used in molecular biology)
- P10-P100: 10-100 μL (molecular biology, biochemistry)
- P100-P1000: 100-1000 μL (general laboratory use)
- P1000-P5000: 1-5 mL (larger sample and reagent handling)
Single-Channel vs. Multichannel:
- Single-channel micropipettes feature a single tip cone and dispense one sample at a time. They offer maximum flexibility for transferring to varied locations and are essential for applications where multiple different volumes are required from a single source.
- Multichannel micropipettes feature 8, 12, or more tip cones arranged to match microplate well spacing (8 mm for 96-well plates, 4.5 mm for 384-well plates). These pipettes dispense simultaneously to multiple wells, dramatically increasing speed for microplate-based applications.
Calibration and Accuracy:
Micropipettes are subject to rigorous calibration according to ISO 8655 standards. Calibration involves weighing distilled water dispensed by the pipette, then converting mass to volume based on water density at ambient temperature. Laboratories must verify accuracy at multiple points across the volume range (typically at 10%, 50%, and 100% of nominal volume) and document results for traceability.
Volumetric Pipettes
Volumetric pipettes are designed to deliver a single, specific, fixed volume with exceptional accuracy. Unlike variable-volume micropipettes, volumetric pipettes cannot be adjusted to different volumes; each pipette is manufactured and calibrated for a single target volume.
Design and Accuracy:
A volumetric pipette consists of a bulbous chamber (the measuring reservoir), a narrow vertical stem with a single graduation mark at the top, and a tapered delivery tip. The characteristic narrow stem is crucial for accuracy: it enables precise reading of the meniscus (the curved surface of the liquid in the pipette), which is read at eye level to determine if the liquid level matches the calibration mark. This narrow geometry means even small variations in meniscus positioning translate to large differences in reading accuracy, requiring focused technique.
Classification by Type and Class:
Volumetric pipettes are classified by:
- Type (delivery characteristics):
- Type 1: Partially deliverable at all volumes
- Type 2: Nominal value at top, zero at bottom (conventional delivery)
- Type 3: Complete delivery only at nominal volume (blow-out pipette)
- Accuracy Class:
- Class A (highest accuracy): Maximum permissible error of ±0.4% of nominal volume. Examples: 25 mL Class A has error limit of ±0.10 mL. Used in analytical chemistry and pharmaceutical assays.
- Class AS: Maximum permissible error of ±0.4% (identical to Class A)
- Class B (general laboratory): Maximum permissible error of ±0.8% of nominal volume (double Class A tolerance). Suitable for general preparation, less stringent applications.
Advantages of Volumetric Pipettes:
- Exceptional Accuracy: When used correctly, volumetric pipettes achieve accuracy superior to variable-volume micropipettes, with errors typically <0.5% of the target volume.
- Simplicity: With no moving parts beyond the pipette body and tip, volumetric pipettes are durable and require minimal maintenance.
- No Calibration Uncertainty: Each Class A pipette is individually calibrated at the factory and certified. The accuracy is fixed and doesn’t degrade over time (assuming no damage).
Limitations:
- Fixed Volumes Only: A different pipette is required for each volume. Running assays requiring 5, 10, 25, and 50 mL volumes requires four separate pipettes.
- Higher Cost per Volume: Because each pipette is calibrated for a single volume, the cost-per-capability is high compared to adjustable micropipettes.
- Slower Workflow: Selecting and using multiple fixed-volume pipettes is more time-consuming than using a single adjustable pipette.
- Skill-Dependent: Reading the meniscus accurately requires training and attention. Improper meniscus reading is a common source of error with volumetric pipettes.
Volumetric pipettes remain the standard for analytical chemistry, titration, and any application where maximum accuracy is required and fixed volumes are sufficient.
Multichannel Pipettes
Multichannel pipettes represent a specialized adaptation of micropipette design, featuring multiple tip cones (typically 8, 12, or 96 channels) arranged in a linear or grid pattern that matches the geometry of microplates.
Design and Mechanism:
Multichannel pipettes function mechanically like single-channel pipettes but deliver liquid simultaneously through multiple channels. Each channel operates independently, allowing different volumes to be set for each channel (in some models). Electronic multichannel pipettes offer superior performance: each channel can be programmed with independent parameters, and consistency across channels is controlled electronically rather than relying on mechanical precision.
Applications and Benefits:
Multichannel pipettes have revolutionized laboratory throughput in:
- High-Throughput Screening: In pharmaceutical drug discovery, screening thousands of compounds against biological targets requires dispensing reagents to 96-well or 384-well microplates. Multichannel pipettes enable filling entire microplate rows simultaneously.
- ELISA and Immunoassays: Microplate-based antibody assays routinely process 40-96 samples in parallel. Multichannel pipettes are essential for adding reagents to all wells quickly and consistently.
- PCR and qPCR: Master mixes are often distributed to 96-well reaction plates using multichannel pipettes, ensuring consistent template amounts across all reactions.
- Cell Culture and Cloning: When plating large numbers of bacterial or mammalian cell colonies, multichannel pipettes dramatically reduce hands-on time and improve consistency.
Efficiency Gains:
By transferring to 8 wells simultaneously, an 8-channel pipette reduces the number of pipetting actions required to fill a 96-well plate from 96 to 12-a 8-fold reduction in repetitive hand motions. For researchers conducting thousands of pipetting operations daily, this translates directly to reduced RSI risk and improved productivity.
Channel Consistency:
A critical requirement for multichannel pipettes is that all channels deliver the same volume with high precision and minimal variation from channel to channel. Electronic multichannel pipettes typically achieve <1% variation between channels; mechanical multichannel pipettes may show 2-3% variation. ISO 8655:2022 now includes specific guidelines for multichannel pipette tolerance and measurement procedures.
Pipette Tips Explained: Critical Section
Pipette tips are the disposable, removable components that attach to the pipette body and physically contact the liquid being transferred. Despite being consumable items, tips are fundamental to pipetting accuracy and represent a critical interface between the pipette mechanism and the sample.
Why Pipette Tips Matter for Accuracy:
The relationship between pipette and tip is far more complex than it initially appears. The tip is not merely a passive container; it is an integral component of the complete pipetting system. ISO 8655:2022 explicitly states that “pipettes and tips are treated as one system,” reflecting the recognition that accuracy depends on both the pipette instrument and the tip working together as a unit.
Several factors contribute to this system relationship:
- Tip Fit and Seal: A poor fit between tip and pipette creates leakage, causing liquid to escape during aspiration and dispensing. This results in delivering substantially less volume than intended.
- Tip Geometry: The exact diameter and shape of the tip cone determines how efficiently the piston creates pressure to draw liquid and how completely liquid is dispensed. Deviations from specification affect volume accuracy.
- Compliance and Elasticity: Plastic tips deform slightly under the pressure created by the piston. This deformation affects the volume of liquid the tip can hold at a given piston position. Tips from different manufacturers-or even different batches-may have different elasticity, resulting in different delivered volumes.
- Surface Properties: The internal surface finish of the tip affects how liquid clings to the plastic (wetting). Hydrophobic surfaces (water-repelling) promote complete liquid drainage, whereas hydrophilic surfaces (water-attracting) cause liquid to adhere to the walls, resulting in lower delivered volumes.
Filtered vs. Non-Filtered Tips:
- Non-Filtered (Standard) Tips: These are the default choice for most applications. They are simple plastic cones with no internal components, minimizing cost and waste.
- Filtered Tips: These tips feature a hydrophobic filter material (typically a microporous membrane made from polyethylene or polypropylene) positioned at the interface between the tip chamber and the tip cone. The filter serves multiple functions:
- Aerosol and Splash Prevention: Reducing the risk of liquid or aerosols entering the pipette shaft and contaminating the piston.
- Cross-Contamination Prevention: Particularly important when pipetting RNA, DNA, or hazardous materials, the filter acts as a physical barrier.
- Sample Protection: For sensitive samples (cDNA libraries, PCR products), filter tips prevent contamination from pipette shaft biofilm or previous samples.
Filter tips are specifically recommended for:
- RNA and DNA work (preventing RNase and DNase contamination)
- PCR and qPCR applications
- Infectious agent handling
- Radioactive or hazardous material pipetting
- Work with volatile, corrosive, or viscous solutions that might splash or aerosolize
Sterile vs. Non-Sterile Tips:
- Non-Sterile Tips: Suitable for routine chemical analysis, general sample preparation, and applications where microbial sterility is not required. These tips represent the most economical choice.
- Sterile Tips: Pre-sterilized by gamma irradiation and certified free of microorganisms and pyrogens (endotoxins). Available with additional certifications:
- DNase/RNase-free Certification: Sterile tips that have been manufactured and packaged to guarantee absence of DNase and RNase enzymes. Simply autoclaving non-sterile tips does not eliminate nucleases; this requires specialized manufacturing and certification.
- Pyrogen-Free/Endotoxin-Free: Critical for applications in cell culture, immunoassays, and pyrogenicity testing.
- DNA/Human DNA-Free: Certified for forensics, genetic testing, and highly sensitive PCR applications.
The temptation to autoclave non-sterile tips to save costs should be resisted for any application requiring nuclease-free conditions. Autoclaving kills living microorganisms but does not degrade or remove nuclease enzymes, which can remain active and contaminate sensitive assays.
Compatibility Issues:
Pipette tips from different manufacturers vary in fit, wall thickness, and surface properties. The consequence of incompatibility can be:
- Loose fit: Tips fall off during pipetting, causing sample spillage and loss.
- Tight fit: Excessive force is required to attach and eject tips, increasing fatigue and hand strain.
- Accuracy Differences: Even when tips fit adequately, different manufacturers’ tips may deliver different volumes on the same pipette because of geometry and material differences.
Many laboratories unknowingly compromise accuracy by using off-brand or incompatible tips to reduce costs. Cross-manufacturer compatibility is not guaranteed; using tips designed for specific pipette brands ensures optimal performance.
Low-Retention Tips:
Some specialized tip formulations include hydrophobic coatings or shaped geometries designed to minimize liquid retention (carryover) on the tip’s internal surface. When dispensing into low-volume systems (such as 384-well microplates with 10-20 μL per well), standard tips can retain 1-3 μL of liquid even after the intended volume is dispensed. This “dead volume” represents 5-30% of the target volume, severely compromising accuracy. Low-retention tips reduce this carryover to <0.5 μL, substantially improving accuracy.
Common Mistakes Labs Make with Pipette Tips:
- Reusing tips across samples: Tips are designed for single use. Reusing tips introduces cross-contamination and carryover effects, reducing accuracy and introducing biological/chemical contamination.
- Using incompatible tips to save costs: Generic or off-brand tips introduce accuracy variability that often exceeds the cost savings.
- Using non-sterile tips for sensitive applications: A common false economy that results in failed assays.
- Storing tips in humid environments: Plastic tips absorb moisture, which affects their properties and can introduce contamination from water.
- Using damaged or scratched tips: Scratches on the tip interior affect liquid flow and volume accuracy. Tips should be visually inspected before use.
Accuracy, Precision & Calibration
Understanding the distinction between accuracy and precision is fundamental to competent laboratory practice, yet these terms are frequently confused or conflated. While closely related, they represent distinct quality attributes of liquid handling instruments.
Accuracy vs. Precision: Clear Definition:
Accuracy refers to how close the delivered volume is to the intended target volume. It measures systematic error-bias in one direction. For example, if a pipette is set to deliver 100 μL but consistently delivers 102 μL, the pipette has a systematic error (positive bias) of +2%, even if the repeated measurements are consistent.
Precision refers to the reproducibility or repeatability of measurements-how consistent results are when the same operation is repeated. It measures random error (variability from one measurement to the next). For example, a pipette might deliver volumes of 100.1, 99.8, 100.2, and 99.9 μL when set to 100 μL. These measurements are highly precise (low variation, with a coefficient of variation <0.5%), but if the true target is 100 μL, they are highly accurate as well.
A pipette can be precise but inaccurate (consistent 2% overdelivery), accurate but imprecise (individual measurements vary by ±5% around a correct mean), or both accurate and precise (the ideal condition).
ISO 8655 Standard for Pipette Calibration:
ISO 8655 is a comprehensive international standard series governing the calibration, testing, and verification of piston-operated volumetric apparatus, including pipettes. Originally published in 2002, the standard was substantially revised in April 2022 (ISO 8655:2022), incorporating more stringent requirements based on two decades of laboratory experience.
The 2022 Revision Key Changes:
- Expanded Structure: Increased from 6 parts to 9 parts, with dedicated sections for pipettes (Part 2), burettes (Part 3), dilutors (Part 4), and dispensers (Part 5).
- System Approach: Pipettes and tips are now explicitly treated as a complete system, not independent components. This reflects the fact that accuracy depends on both working together.
- Multiple Volume Error Limits: The previous standard specified error tolerance only at the nominal (maximum) volume. The revised standard specifies tolerance at 10%, 50%, and 100% of nominal volume, acknowledging that accuracy varies across the pipette’s operating range.
- Stricter Environmental Control: Calibration procedures now specify precise ranges for temperature (20°C ± 2°C), humidity (45-75%), and atmospheric pressure, recognizing that these variables affect measurement validity.
- Enhanced Balance Requirements: Gravimetric calibration now requires higher-precision balances:
- 6-place balance (resolution 0.001 mg) for pipettes <20 μL
- 5-place balance (resolution 0.01 mg) for 20-199 μL pipettes
- 4-place balance (resolution 0.1 mg) for larger volumes
Gravimetric Calibration Method (ISO 8655-6):
The gravimetric method is the international reference standard for pipette calibration and verification. It is the most widely accepted and reproducible approach.
Principle: Distilled water of known density is dispensed into a pre-weighed container and weighed. The mass of water dispensed is converted to volume using the relationship: Volume (mL) = Mass (g) / Density of water at reference temperature. Since water density varies with temperature, calibration is performed at a reference temperature (typically 20°C), and density is corrected accordingly.
Calibration Procedure:
- Preparation: The balance is calibrated and verified. Environmental conditions (temperature, humidity) are recorded and confirmed to be within specification ranges. The pipette is checked for visible damage.
- Volume Selection: At minimum, three representative volumes are selected for testing:
- The nominal (maximum) volume
- 50% of the nominal volume
- The maximum of either 10% of nominal volume or the lower limit of the useful volume range
For variable-volume pipettes used across a wide range (e.g., P1-P100), additional test points may be selected.
- Tip Equilibration: The selected tip type is attached to the pipette and used for all calibration measurements. The tip must equilibrate to the ambient temperature and humidity, typically requiring 30-60 minutes.
- Prewetting: The tip is wetted by drawing in and expelling the target liquid 3-5 times. This equilibrates the temperature and ensures accurate measurements by reducing evaporation effects.
- Dispensing and Weighing: For each test volume:
- A pre-weighed container is placed on the analytical balance
- The pipette (with attached tip) aspirates the target volume
- The pipette dispenses the volume into the container
- The container is weighed, and the mass of liquid is recorded
- This procedure is repeated a minimum of 10 times per volume, then at least once more at maximum and minimum volumes
- Data Analysis:
- For each series of measurements, the mean volume and standard deviation are calculated
- Accuracy is assessed by comparing the mean volume to the nominal (expected) volume; the difference is the systematic error
- Precision is assessed by calculating the coefficient of variation (CV) = (Standard Deviation / Mean) × 100%; this represents random error
- Both are compared against tolerance limits specified in ISO 8655-2 for the pipette’s nominal volume and class
- Documentation: All results are recorded on a calibration certificate, including:
- Pipette model and serial number
- Date of calibration
- Environmental conditions during calibration
- Test volumes, mean values, and standard deviations
- Pass/Fail determination for each volume
- Signature and certification by the accredited laboratory
- The next recommended calibration date
Acceptable Tolerances (ISO 8655-2 for Single-Channel Pipettes):
For example, a 1000 μL Class A micropipette has the following maximum permissible errors:
- At 10% of nominal (100 μL): ± 10% = ±10 μL
- At 50% of nominal (500 μL): ± 2% = ±10 μL
- At 100% of nominal (1000 μL): ± 1% = ±10 μL
Note that the absolute error limit (±10 μL) remains constant across this range, while the percentage error improves at higher volumes. This reflects the physical reality that absolute measurement uncertainty is relatively fixed for air-displacement pipettes but becomes a smaller percentage of larger volumes.
Consequences of Poor Calibration:
Laboratories that neglect calibration or use uncalibrated pipettes risk:
- Invalid Assay Results: Especially in quantitative analysis, poor calibration introduces systematic bias that invalidates results.
- Failed Regulatory Audits: Accreditation bodies (CAP, CLIA, ISO 17025) require documented evidence of regular pipette calibration. Failure to calibrate is a major audit finding.
- Reduced Statistical Power: Increased random error from poorly calibrated pipettes requires larger sample sizes to achieve statistical significance.
- Compromised Reproducibility: Results cannot be reliably reproduced in other laboratories or when using different pipettes.
How Often Calibration Is Needed:
ISO 8655 and most laboratory accreditation standards recommend calibration:
- Initial Calibration: Before first use or when initially placed in service
- Periodic Calibration: Typically annually for properly maintained pipettes in routine use
- As-Needed Calibration: After visible damage, drops, or suspected malfunction; after extensive use in demanding applications; whenever accuracy seems questionable
- Pre-Use Verification: Many labs perform simplified verification (dispensing water and comparing weight to expected mass) monthly or quarterly to flag instruments requiring recalibration before damage accumulates
Large laboratories often use automated balance systems that perform gravimetric calibration of multiple pipettes in sequence, enabling more frequent testing while reducing technician time.
Ergonomics and User Safety
While pipettes are not typically considered hazardous equipment in the sense of involving extreme temperatures, high pressures, or toxic exposure, they represent one of the most significant occupational health threats in laboratory settings due to repetitive strain injury (RSI). This chronic injury category represents a major occupational health burden that is entirely preventable through proper ergonomic design and technique.
Repetitive Strain Injury (RSI) and Laboratory Workers:
RSI encompasses a group of musculoskeletal disorders characterized by pain, inflammation, or other dysfunction in muscles, tendons, and nerves resulting from repetitive motions or sustained awkward postures. In laboratory settings, pipetting is the primary source of RSI, with research documenting that:
- Approximately 40% of laboratory professionals report pipetting-related pain or dysfunction in the hands, wrists, arms, shoulders, or back.
- Nearly 90% of researchers who pipette more than one hour daily experience hand or elbow pain or dysfunction.
- Common diagnoses include carpal tunnel syndrome, tendonitis, tenosynovitis (inflammation of the tendon sheath), and epicondylitis (tennis elbow).
Anatomical Basis: When a hand and wrist are positioned in slight extension (bent backward) and a pinching force is applied repeatedly thousands of times daily, the flexor and extensor tendons of the forearm are subjected to continuous stress. These tendons must glide through tight tunnels (carpal tunnel, cubital tunnel) to reach the fingers. With repeated stress, inflammation develops, swelling increases pressure within these fixed tunnels, compressing nerves and causing pain, tingling, and eventual permanent nerve damage if untreated.
Three Major Risk Factors for RSI in Pipetting:
- Posture: Prolonged static positioning of the hand and wrist in extension (fingers pointing downward while holding the pipette) is inherently stressful. A hunched posture over the lab bench compounds the problem by adding shoulder and neck strain. Proper ergonomic posture requires:
- The shoulder in neutral position (not elevated or retracted)
- The elbow at approximately 90 degrees
- The wrist as neutral as possible (not bent backward or forward)
- The work surface at a height allowing the forearms to be parallel to the ground
- Force: The force required to operate a pipette is application-dependent:
- Tip Attachment/Ejection: This is the most force-intensive step. Stiff or poorly designed tip attachment mechanisms can require 10-20 pounds of force, which over hundreds of repetitions causes cumulative strain. Electronic tip ejection (controlled by a motor) reduces this to the equivalent of a keyboard keystroke (<5 ounces of force).
- Plunger Operation: Manual pipettes with stiff plunger mechanisms require sustained gripping force. Conversely, pipettes with smooth, low-friction plungers minimize required force.
- Grip Strength: The overall grip force required to hold the pipette during operation varies by design. Poorly ergonomic handles with inadequate finger support increase required grip force.
- Repetition: The sheer frequency of pipetting motions is the defining factor. A researcher conducting high-throughput screening might perform 10,000-50,000 pipetting cycles per day. Each cycle involves:
- Grasping the pipette (gripping force)
- Positioning the tip (wrist/arm positioning)
- Pressing the plunger (finger extension/flexion)
- Ejecting the tip (force application)
- Reaching for the next tip (shoulder/arm motion)
- Repositioning for the next aspirate or dispense
When multiplied by thousands, these seemingly minor stresses accumulate into significant musculoskeletal load.
Impact on Lab Productivity and Quality:
Contrary to the assumption that ergonomic improvements are “nice to have” quality-of-life issues, RSI directly impacts laboratory performance:
- Reduced Accuracy: Pain and fatigue compromise fine motor control, introducing pipetting errors.
- Decreased Throughput: Researchers experiencing RSI work more slowly and require frequent breaks.
- Unscheduled Absences: Chronic RSI often leads to medical leave or reduced-hours arrangements, disrupting laboratory operations.
- Staff Turnover: Severe RSI can force talented researchers out of laboratory work entirely, increasing recruitment and training costs.
How Ergonomic Design Reduces RSI:
Manufacturers and laboratory designers have developed pipette features specifically to reduce RSI risk:
- Electronic Pipetting: By replacing manual plunger operation with motor-controlled dispensing, electronic pipettes reduce repetitive hand motion and force. Researchers using electronic multichannel pipettes report 50-70% less hand pain than those using manual single-channel pipettes.
- Low Operating Force Plungers: Smooth, low-friction plunger mechanisms reduce the grip force required. Comparing a manual P1000 with a stiff plunger (requiring ~15 ounces of force per cycle) to an ergonomic model (requiring ~8 ounces per cycle) and multiplying by 5,000 daily cycles demonstrates the cumulative benefit.
- Ergonomic Grip Design: Properly contoured handles with padded surfaces and finger hooks distribute grip forces more evenly and reduce overall required force. The best designs support both right- and left-handed use.
- Lightweight Construction: Heavy pipettes (>50 grams) increase fatigue during extended use. Modern ergonomic designs minimize weight while maintaining durability.
- Electronic Tip Ejection: Motorized ejection eliminates the forceful button press required with manual ejection, replacing it with the force equivalent to pressing a computer key (~2 ounces).
- Multichannel Pipettes: By reducing the number of pipetting actions from 96 to 12 (when using 8-channel pipettes on 96-well plates), multichannel models proportionally reduce repetitive strain. An 8-channel pipette used on a 96-well plate reduces the number of pipetting cycles from 96 to 12-an 8-fold reduction in wrist and hand stress.
Proper Pipetting Technique to Prevent RSI:
Regardless of pipette design, proper technique is essential:
- Maintain neutral wrist position: Avoid bending the wrist backward (extension) while pipetting. A vertical or slightly forward wrist angle reduces tendon stress.
- Position arms correctly: Keep upper arms near the body; avoid reaching across work surfaces. Position the lab bench at a height allowing forearms to be roughly parallel to the ground.
- Use a light grip: Grip force should be only sufficient to maintain control. A death-grip on the pipette introduces unnecessary strain. The pipette should be balanced in the hand, not gripped tightly.
- Take regular breaks: Every 20-30 minutes of continuous pipetting, take a 2-5 minute break to allow muscles and tendons to recover.
- Perform hand and arm stretches: Regular stretching maintains flexibility and reduces muscle tension. Specific stretches for the forearm flexors and extensors are particularly beneficial for pipetting tasks.
- Vary tasks: If possible, alternate pipetting with other laboratory tasks to reduce continuous stress on the same muscle groups.
- Use two-handed pipetting when possible: For bulky or heavy pipettes, using both hands distributes load and reduces strain on one side.
How to Choose the Right Pipette for Your Laboratory
Selecting appropriate pipette instrumentation requires systematic consideration of multiple factors. A decision framework that addresses these factors sequentially leads to optimal selections that balance accuracy requirements, application needs, budget constraints, and user ergonomics.
Decision Framework:
1. Define the Volume Range Required:
The first criterion is identifying the range of liquid volumes your laboratory routinely pipettes. Different pipettes excel at different volume ranges:
- Ultra-low volumes (0.5-10 μL): Specialized ultra-precision pipettes with low air dead volume
- Low volumes (10-100 μL): Standard micropipettes used in molecular biology
- Medium volumes (100-1000 μL): General laboratory pipettes
- Large volumes (1-5 mL): Macropipettes for bulk reagents and media
- Very large volumes (>5 mL): Graduated pipettes or motorized dispensers
Selecting a pipette whose range encompasses your application improves accuracy. Pipetting at the minimum or maximum of a pipette’s range introduces greater error than pipetting near the mid-range. For applications requiring volumes of 50-500 μL, a P1-P100 micropipette would be inappropriate (using 500 μL on a P1-P100 maximum of 100 μL is impossible); conversely, using a P1000 for a 50 μL volume would be suboptimal (50 μL is near the minimum of a P1-P1000, where accuracy is poorest).
2. Assess Accuracy and Precision Requirements:
Different applications demand different levels of accuracy:
- High-accuracy applications (analytical chemistry, titration, pharmaceutical assays, clinical diagnostics): Class A volumetric pipettes or electronic micropipettes with CV <0.5%
- Moderate-accuracy applications (general biochemistry, solution preparation): Electronic or well-maintained manual micropipettes with CV 0.5-1.5%
- Lower-accuracy applications (rough reagent dispensing, waste disposal): Basic manual pipettes or graduated pipettes
3. Evaluate the Application Type and Workflow:
Different workflows benefit from different pipette types:
- Single-sample pipetting (preparing individual standards, processing discrete samples): Single-channel pipettes provide maximum flexibility
- Multiple-sample processing in parallel (ELISA plates, PCR plate setup): Multichannel pipettes dramatically improve efficiency
- Precise fixed-volume transfers (chromatography sample injection, standard preparation): Volumetric pipettes or electronic single-channel with fixed volume buttons
- High-throughput screening (96-well or 384-well plate processing): Electronic 12-channel or 96-channel pipettes
4. Consider Budget and Cost of Operation:
Initial purchase cost represents only one dimension of total cost of ownership:
- Purchase Price: Electronic pipettes ($2,000-$8,000 per unit) vs. manual pipettes ($200-$1,500 per unit)
- Tip Costs: Bulk-packaged tips cost approximately $0.01-$0.05 per tip; filter tips or sterile tips cost $0.03-$0.10 per tip. For a laboratory conducting 100,000 pipetting operations monthly, tip costs are substantial ($1,000-$10,000 annually)
- Maintenance: Electronic pipettes require annual servicing ($200-$500 per instrument); manual pipettes require minimal maintenance
- Calibration: Annual calibration costs $100-$300 per instrument at external calibration service or can be performed in-house with proper equipment
- Longevity: Manual pipettes often function adequately for 10-15 years; electronic pipettes typically have 7-10 year effective life (though modern models are increasingly durable)
A comprehensive cost-benefit analysis often reveals that electronic pipettes, despite higher initial cost, deliver better economics when labor costs and improved productivity are considered.
5. Differentiate Between Beginner and Professional Labs:
Beginner Laboratories (teaching labs, startup biotech firms, or temporary research):
- Prioritize cost-effectiveness and ease of use
- Invest in basic manual single-channel pipettes with established reliability records
- Focus on proper training and technique before upgrading to advanced instruments
- A basic kit of manual pipettes (P10-P100 for molecular work or P100-P1000 for general work) represents a solid starting point
Professional Research and Clinical Laboratories:
- Prioritize accuracy, throughput, and ergonomics
- Invest in electronic multichannel pipettes for high-throughput applications
- Implement systematic calibration and maintenance programs
- For high-accuracy analytical work, combine electronic micropipettes with volumetric pipettes for critical transfers
- Consider automated liquid handling platforms when pipetting exceeds 10,000 actions daily
Sample Selection Guidance:
For a typical molecular biology laboratory:
- Molecular biology pipette kit (general use): P2-P20 (ultra-precision for qPCR), P20-P200 (PCR, DNA work), P200-P1000 (buffer and media dispensing). Electronic models are preferred; multichannel 8-channel P20-P200 and P200-P1000 for 96-well plate work.
- Additional recommendation: A Class A 1 mL volumetric pipette for preparing critical standards.
For a clinical laboratory:
- High-throughput microplate orientation: Electronic 12-channel P2-P20 and P20-P200 pipettes for microplate assays; manual P1-P10 for ultra-precision clinical measurements
- Single-channel electronic P2-P200 for general use
- Systematic calibration program with quarterly verification
For a pharmaceutical quality control lab:
- Electronic P2-P20, P20-P200, and P200-P1000 single-channel pipettes
- Class A volumetric pipettes (1, 2, 5, 10, 25, 50 mL) for reference standard preparation
- Documented calibration every 6 months with certificates for regulatory compliance
Common Pipetting Errors and How to Avoid Them
Even with properly functioning, well-calibrated pipettes, systematic errors can arise from improper technique or environmental factors. Understanding common error sources enables researchers to implement corrective measures and improve accuracy.
Error 1: Angle of Tip Immersion
The Problem: When a pipette tip is immersed at an angle significantly different from 90 degrees (perpendicular) to the liquid surface, the amount of liquid aspirated can deviate substantially from the intended volume.
Mechanism: An angled tip causes uneven pressure distribution on the liquid surface, resulting in incomplete seal or variable fill. Additionally, the effective depth of tip immersion varies with angle, affecting the amount of liquid aspirated. Studies show that aspirating at 45 degrees (instead of 90 degrees) can result in 10-20% volume errors.
Prevention:
- Always insert the tip vertically into the liquid, ensuring a 90-degree angle with the liquid surface
- Use microplates with wells positioned at a consistent height to maintain consistent insertion angle
- Position your hand and pipette naturally; this typically results in the correct vertical orientation
- For viscous liquids (glycerol, oils), ensure the tip is fully immersed; for aqueous solutions, immersion of 2-3 mm below the surface is typically sufficient
Error 2: Aspiration and Dispensing Speed
The Problem: The speed at which the plunger is pressed and released affects the volume of air cushion changes and can introduce errors, particularly with challenging liquids or small volumes.
Mechanism: Rapid plunger movement can cause:
- Splashing of liquid against the pipette shaft (especially with aqueous solutions), creating aerosols that escape the tip
- Incomplete filling when the aspirate speed exceeds the rate at which liquid can flow into the tip
- Turbulence and mixing, which can affect the physical properties of the liquid (relevant for volatile or foaming solutions)
Slow plunger movement can cause:
- Evaporation effects, particularly important for volatile solvents or when pipetting at temperature extremes
- Excessive time for temperature equilibration effects to occur
Prevention:
- Use smooth, controlled plunger motion at moderate speed
- Electronic pipettes programmed with appropriate speed profiles are superior to manual operation
- For volatile liquids, minimize time from aspiration to dispensing
- For viscous or high-density liquids, use slower speeds to ensure complete filling
- Avoid aggressive plunger motion; the motion should be deliberate and controlled, not jerky or forceful
Error 3: Insufficient Prewetting
The Problem: When a fresh tip is used without first drawing in and expelling the liquid several times, the temperature and humidity conditions inside the tip may differ from the target liquid, resulting in systematic error.
Mechanism: Prewetting addresses two issues:
- Temperature Equilibration: If a room-temperature pipette tip is used to aspirate a cold liquid (restriction enzyme buffer at 4°C), the air cushion inside the tip initially contracts as temperature drops, resulting in reduced volume delivery. Prewetting brings the tip to liquid temperature, eliminating this source of error.
- Humidity Equilibration: Plastic tips absorb or release moisture depending on ambient humidity and the hygroscopic properties of the liquid being pipetted.
Studies show that failure to prewet can introduce 2-5% systematic error, particularly with small volumes or temperature-sensitive applications.
Prevention:
- Before beginning a series of pipetting operations, aspirate and dispense the target liquid 3-5 times
- Electronic pipettes can be programmed to automatically include prewetting steps
- For cold reagents (4°C storage), prewetting is particularly critical
- For room-temperature solutions, prewetting is still recommended but somewhat less critical than for temperature-challenged liquids
Error 4: Temperature Effects and Thermal Disequilibrium
The Problem: When pipettes are used to transfer liquids at temperatures significantly different from ambient room temperature, substantial volume errors can occur. This is particularly pronounced with small-volume pipettes and air-displacement instruments.
Mechanism: Air-displacement pipettes function by maintaining a small air cushion between the piston and the liquid. This air expands or contracts with temperature changes. When a cold liquid (e.g., restriction enzyme buffer at 4°C) is aspirated into a room-temperature pipette:
- The air inside the pipette tip is compressed as the piston moves to draw in liquid
- Upon contact with the cold liquid, the compressed air rapidly cools and contracts
- This contraction allows additional liquid to be drawn into the tip, resulting in over-delivery
Conversely, with hot liquids (e.g., PCR reaction master mixes at 55°C):
- The air in the tip expands, pushing liquid out
- This results in under-delivery
The magnitude of error depends on:
- Volume of pipette: Smaller pipettes with proportionally larger air cushions show greater error (up to 37% error when using a 2 μL pipette at minimum volume with extreme temperature differences)
- Temperature differential: Greater differences between pipette and liquid temperature produce greater errors
- Volume being dispensed: Pipetting at the minimum volume setting (where the air cushion is largest relative to liquid volume) produces greater errors than pipetting at the maximum volume
Prevention:
- Optimal approach: Allow liquids to reach room temperature before pipetting when possible. For restriction enzyme digestions or PCR setup where room-temperature incubation is not appropriate, this may require accepting greater error margins.
- Use prewetting: The prewetting procedure helps equilibrate the air cushion temperature
- Minimize dead air volume: Pipette at volumes closer to the maximum of the pipette’s range. Using a P200 to deliver 200 μL (rather than a P10 to deliver 10 μL) dramatically reduces relative error.
- Select positive-displacement pipettes: For applications requiring repeated transfer of non-ambient temperature liquids, positive-displacement pipettes (which eliminate the air cushion) provide substantially better accuracy
- Allow additional equilibration time: Allow pipette and tip to be in contact with the cold/hot environment for 1-2 minutes before beginning pipetting operations
Error 5: Tip Immersion Depth
The Problem: The depth to which the pipette tip is immersed in the liquid affects the volume aspirated, particularly for viscous or non-aqueous solutions.
Mechanism: For aqueous solutions with standard air-displacement pipettes, immersion depth is relatively non-critical for reasonably shallow immersions (2-5 mm). However, for viscous or surface-tension-active liquids:
- Shallow immersion may result in incomplete filling, particularly if the liquid does not flow readily into the tip
- Deep immersion (>5-10 mm) introduces additional hydrostatic pressure from the column of liquid above the tip, which can affect the measured volume
Prevention:
- For aqueous solutions: Immerse 2-3 mm below the liquid surface
- For viscous solutions: Full or near-full immersion may be necessary for complete filling
- For foaming or volatile liquids: Shallow immersion (just below surface) minimizes aerosolization
- Avoid dragging the tip along the bottom of wells or containers; this can create irregular pressure conditions
Error 6: Viscosity and Liquid Properties
The Problem: Liquids with properties significantly different from water (viscosity, volatility, surface tension, density) behave differently in pipettes and can introduce substantial errors in air-displacement instruments.
Mechanism:
- High viscosity (glycerol, oils, protein solutions): Thick liquids flow more slowly into the pipette tip and cling to the plastic, reducing the volume that drains from the tip after dispensing. This results in under-delivery.
- Low viscosity (methanol, acetone, volatile organic solvents): These flow readily into the tip but evaporate from the tip’s internal surface, reducing delivered volume. Evaporative losses can be 5-10% for volatile solvents at room temperature.
- High surface tension: Liquids with high surface tension (like glycerol or serum) tend to cling to plastic surfaces, reducing drainage from the tip.
- Non-aqueous density: Non-aqueous solutions have different density than water. Gravimetric calibration (which assumes water density) introduces error when these liquids are pipetted.
Prevention:
- Use positive-displacement pipettes for viscous or challenging liquids. These pipettes, with direct piston-to-liquid contact, are unaffected by viscosity changes and experience much smaller evaporation effects.
- For aqueous solutions with additives, perform calibration with the specific solution (not just water) if precision is critical. This accounts for density and viscosity effects.
- For volatile solvents: Minimize time from aspiration to dispensing. Consider using closed pipette systems or positive-displacement pipettes.
- For viscous solutions: Dispense slowly to allow complete drainage, and consider using wide-bore tips that facilitate drainage.
- Perform pilot testing: Before committing to critical assays involving unusual liquids, perform calibration or verification tests with the actual liquid to quantify any systematic errors.
Error 7: Contamination and Cross-Contamination
The Problem: Residual liquid or particulates from one sample can contaminate the next sample if proper pipetting technique and hygiene are not maintained.
Mechanism:
- Reusing pipette tips introduces direct carryover from one sample to the next
- Inadequate cleaning of the pipette shaft allows biofilm and previous sample residues to leach into new samples (particularly important for sterile work and nucleic acid applications)
- Aerosols created during pipetting can settle on pipette exteriors and subsequently enter new samples
- Improper hand hygiene during high-throughput work introduces contamination from previous samples on gloved hands
Prevention:
- Use fresh tips for every sample: This is the most critical factor. Never reuse tips across different samples.
- Use filtered tips for sensitive applications: Filtered tips prevent aerosols and splash-back from entering the pipette shaft.
- Maintain pipette cleanliness: Electronic pipettes should be regularly wiped with appropriate disinfectants. Shafts should be inspected for visible residue.
- Change gloves frequently: Particularly between different samples or between handling reagents and samples.
- Work in properly maintained fume hoods or BSCs when handling hazardous or infectious materials.
- Organize workflow: Arrange samples and reagents to avoid backward steps (moving from contaminated to clean areas).
Manual vs. Electronic Pipettes – Comparison Table
| Feature | Manual Pipettes | Electronic Pipettes |
|---|---|---|
| Operating Principle | User operates plunger by hand; direct mechanical operation | Motorized piston control; programmed dispensing profiles |
| Accuracy (Coefficient of Variation) | Typically 1-3% CV; operator-dependent | Typically <0.5% CV; consistent across operators |
| Precision | Variable, dependent on operator technique and experience | Highly consistent; minimal batch-to-batch variation |
| Initial Purchase Cost | $200-$1,500 per unit (single-channel) | $2,000-$8,000 per unit (single-channel); $4,000-$15,000+ (multichannel) |
| Maintenance Costs | Minimal; simple mechanical service | $200-$500 annually for calibration/service |
| Operating Force Required | 10-20 lbf for tip ejection; variable manual plunger force | <2 lbf (equivalent to keyboard keystroke) for tip ejection; consistent motor operation |
| Ergonomics & RSI Risk | Higher risk; repetitive plunger operation and gripping cause strain | Lower risk; motor-driven operation and programmable features reduce strain |
| Throughput | Lower; limited by manual operation speed | Higher; electronic features and multichannel models increase speed |
| Multichannel Capability | Available but less common; mechanical consistency challenging | Widely available; 8-, 12-, or 96-channel formats; electronic consistency ensures accuracy across channels |
| Temperature Equilibration | Manual prewetting required | Can be programmed to include automatic prewetting steps |
| Learning Curve | Minimal; direct mechanical feedback helps users develop technique | Steeper initially; more features to understand, but superior reproducibility once mastered |
| Versatility | Limited by mechanical design; one dispense profile | Programmable; customizable for different liquids and applications |
| Lifespan | 10-15 years typical | 7-10 years typical (improving with newer designs) |
| Ideal Applications | Single-sample precise pipetting; analytical chemistry; undergraduate labs | High-throughput screening; clinical diagnostics; any application prioritizing accuracy and consistency |
Frequently Asked Questions (FAQ)
Q1: What is the best pipette for beginners?
A: For beginners, a manual single-channel pipette in the 20-200 μL or 100-1000 μL range is ideal. These pipettes are affordable ($300-$800), mechanically simple, and provide direct feedback that helps users develop proper technique. Select a pipette from a reputable manufacturer (Gilson Pipetman, Eppendorf, Rainin, or equivalent) with established documentation and reliable performance. The emphasis should be on proper training: demonstrating correct holding position, plunger operation, and tip immersion before independent use. Once proper technique is established and volume requirements grow, upgrading to electronic or multichannel pipettes is appropriate.
Q2: Do people still use mouth pipetting?
A: No, mouth pipetting is prohibited in all modern accredited laboratories and has been banned by OSHA and the NIH since the 1970s-1980s. Historically, mouth pipetting was responsible for approximately 40% of laboratory-acquired infections (particularly between 1915-1963) and remains implicated in documented cases of typhoid fever, plague, and hepatitis from laboratory exposures. Modern mechanical pipetting devices have eliminated any rational justification for oral pipetting. Any laboratory permitting mouth pipetting is in severe violation of laboratory safety standards and should be reported to occupational safety authorities.
Q3: Which pipette brand is best?
A: Multiple manufacturers produce quality pipettes meeting ISO 8655 standards, including Gilson (Pipetman line), Eppendorf, Rainin (Liquidator and Futura brands), Thermo Fisher (F-series), Sartorius, Mettler Toledo, Integra, and others. Rather than advocating for a specific brand, the selection should be based on:
- Meeting accuracy and performance specifications for your application
- Ergonomic design features appropriate for your user population
- Availability of qualified service and repair locally
- Compatibility with your existing laboratory infrastructure (tips, storage racks, automated systems)
- Total cost of ownership, including tips, maintenance, and calibration
Conduct a small evaluation by borrowing demo units from manufacturers and having your actual users test them for comfort and performance before making a large institutional purchase.
Q4: What is the difference between F1 and F2 pipettes (or Class A vs. Class B volumetric pipettes)?
A: F1 pipettes (equivalent to ISO Class A volumetric pipettes) are manufactured to the highest accuracy standards, with maximum permissible errors of ±0.4% of the nominal volume. These are used for precise analytical work, preparation of standards, and any application where high accuracy is required.
F2 pipettes (equivalent to ISO Class B) have twice the tolerance of F1 pipettes (±0.8% maximum error) and are suitable for general laboratory work where high precision is not critical.
The difference in cost is modest (typically 20-30% more for F1 vs. F2), making F1 pipettes the preferred choice for most analytical applications. F2 pipettes are appropriate only when preparing rough solutions or dispensing into non-critical processes.
Q5: How do ergonomic pipettes improve accuracy & efficiency?
A: Ergonomic pipettes improve both accuracy and efficiency through several mechanisms:
- Reduced operator fatigue: By requiring less gripping force and enabling faster operation, ergonomic features reduce hand and arm fatigue that otherwise compromises fine motor control and technique, thereby improving accuracy.
- Higher throughput: Electronic pipettes and multichannel models process samples much faster, enabling researchers to handle more samples in the same time, increasing efficiency.
- Consistent performance: Ergonomic electronic pipettes deliver consistent volumes across different operators and shifts, improving accuracy by reducing operator-dependent variability.
- Reduced errors from fatigue-related mistakes: Studies show that researchers make more pipetting errors as they fatigue; ergonomic instruments prevent this fatigue-related degradation.
- Improved retention of skilled workers: By preventing RSI, ergonomic instruments help retain experienced researchers, maintaining high-quality work and institutional knowledge.
Summary & Key Takeaways
Pipettes are precision instruments whose selection, operation, and maintenance directly impact laboratory accuracy, regulatory compliance, and worker health. Key points for laboratory professionals:
Type Selection:
- Manual single-channel pipettes are appropriate for lower-throughput analytical work and training, but electronic multichannel pipettes are increasingly standard for professional laboratories due to superior accuracy and throughput.
- Micropipettes (variable-volume) suit most general applications; volumetric (fixed-volume) pipettes are essential for maximum-accuracy analytical work.
- Multichannel pipettes are indispensable for microplate-based applications.
Accuracy and Calibration:
- ISO 8655:2022 is the international standard governing pipette calibration and testing; compliance is required for accredited laboratories.
- Gravimetric calibration is the reference method, involving weighing water dispensed by the pipette.
- Regular calibration (annually minimum, semi-annually for high-use instruments) is essential for regulatory compliance and maintaining accuracy.
Pipette Tips:
- Tips are integral to the pipetting system; compatibility with the specific pipette is critical.
- Sterile and filtered tips are not optional for sensitive applications but essential; RNase/DNase-free certification is required for nucleic acid work.
- Single-use tips prevent cross-contamination and should never be reused.
Ergonomics and Safety:
- Repetitive strain injury is common among laboratory workers but entirely preventable through proper ergonomic design and technique.
- Electronic pipettes with motor-driven operation reduce RSI risk by 50-70% compared to manual pipettes.
- Proper technique (neutral wrist posture, controlled motion, frequent breaks) is essential even with ergonomic instruments.
Error Prevention:
- Temperature equilibration (prewetting), proper tip immersion angle, and appropriate speed are critical for accuracy.
- Positive-displacement pipettes should be used for viscous or volatile liquids where air-displacement instruments show poor performance.
- Maintenance of environmental conditions (temperature, humidity) during storage and use affects accuracy.
Professional Best Practices:
- Implement systematic pipette calibration and maintenance programs.
- Provide comprehensive training to all operators on proper technique and ergonomic positioning.
- Evaluate and upgrade pipettes based on application requirements, not solely on initial purchase cost.
- Document all pipetting procedures and maintain traceability for regulatory and quality purposes.
Laboratory pipetting represents a small but essential fraction of experimental work. Investing in proper instruments, training, calibration, and technique yields disproportionate benefits: improved accuracy, enhanced reproducibility, regulatory compliance, reduced errors, and most importantly, protection of the health and productivity of laboratory professionals who deserve to work safely and effectively.