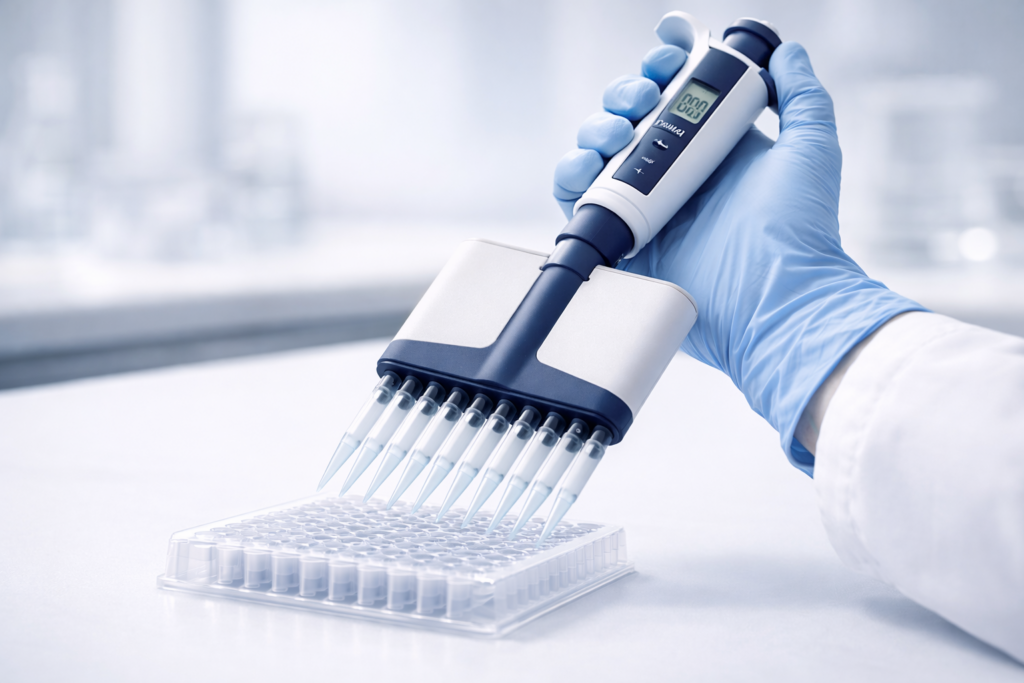
A multi-channel electronic pipette is a motorized liquid-handling device that aspirates and dispenses multiple samples simultaneously using 8, 12, or more channels aligned with microplate formats. Unlike manual multichannel pipettes that rely entirely on thumb force, electronic variants use programmable piston motors to deliver consistent volumes across all channels with reduced user variability. Labs adopt electronic multichannel pipettes to accelerate high-throughput workflows, minimize repetitive strain injuries, and achieve better accuracy on plate-based assays-whether PCR setup, ELISA screening, or cell-based assays. This guide walks you through how they work, when they outperform alternatives, and how to choose the right one for your lab’s needs.
What Is a Multi Channel Electronic Pipette?
An electronic multichannel pipette is a motor-driven instrument that delivers programmable liquid volumes through multiple independently actuated tips in a single operation. The key distinction from manual multichannel pipettes is that electronic models remove the user’s thumb from the aspiration-dispensing cycle: a microprocessor controls piston speed, dwell time, and multi-dispense sequences. Typical channels come in 8 (standard 96-well row), 12 (standard 384-well column), or 16/24 for ultra-high-throughput applications.
Common use cases:
- Filling 96-well PCR plates with master mix or individual samples
- Washing steps in ELISA protocols across multiple rows simultaneously
- Serial dilutions across plates with programmable decrements
- Repeated dispensing of identical volumes (e.g., substrate or stop reagent in colorimetric assays)
How It Works (Simple but Accurate)
Electronic multichannel pipettes operate via a brushless motor that drives a single piston rod connected to all channels in parallel. The motor’s speed, direction, and dwell time are regulated by firmware responding to button input and stored protocols. This architecture differs critically from manual models, where each thumb stroke produces variable force, speed, and acceleration.
Why electronic control reduces user variability:
- Consistent piston velocity: Motor delivers the same acceleration profile on every stroke, regardless of operator fatigue.
- Programmable speed control: Slow aspiration minimizes bubble formation; fast dispensing accelerates workflows.
- Dwell time precision: The instrument pauses at aspiration or dispensing to equilibrate pressure, reducing drips and splashing.
- Multi-dispense without user reset: The pipette can dispense identical sub-volumes to multiple destinations from a single aspiration-impossible without programming.
What still depends on technique:
- Tip sealing: Even with electronic precision, poor tip fit or bent tips will leak. Uniform vertical insertion pressure across all eight channels is essential.
- Immersion depth: Consistent depth (typically 2–3 mm) across wells prevents bubbles and variable backpressure.
- Sample properties: Viscosity, volatility, surface tension, and protein content require adjusted speed settings and possible reverse pipetting-software can’t compensate for poor technique.
- Temperature: Density changes in volatile or cold-start samples affect calibrated volumes.
8 vs 12 Channels (and When 16/24 Matters)
Channel count determines which plate formats and workflow patterns the pipette suits best. Below is a practical breakdown:
| Workflow | Best Channel Count | Typical Volume Range | Notes |
|---|---|---|---|
| PCR strip setup or 8-strip tubes | 8-channel | 1–200 µL | Matches single row of 96-well plate; ideal for qPCR prep |
| 96-well plate (standard full plate fill) | 8-channel | 5–300 µL | Requires 12 operations (8 rows × 1 column pass); fastest manual option for standard plates |
| 384-well plate columns | 12-channel | 2–50 µL | Fills 384 plate in 32 operations (32 columns × 1 pass); reduces plate-to-plate variability |
| High-throughput screening (full plate simultaneous) | 16/24 or 96-channel | varies | Specialized systems; rarely used in routine labs; higher cost and complexity |
| Irregular patterns (scattered wells, mixed tubes/plates) | Single-channel | 0.5–10,000 µL | Multichannel inflexible for non-aligned targets; single-channel more practical |
Key trade-off: 8-channel is versatile for standard 96-well workflows but requires more passes. 12-channel is faster for 384-well plates but less compatible with manual 96-well work. Choose 8 if your lab primarily uses 96-well; choose 12 if 384-well or PCR-strip-to-384 conversion is common.
Electronic Multichannel vs Manual Multichannel vs Single-Channel
Each tool excels in different scenarios. Understanding when to reach for each prevents over-investing in electronics you don’t need and under-investing when they save hours.
| Scenario | Best Tool | Why |
|---|---|---|
| Filling 96 wells with one reagent, all identical volumes | Electronic 8-channel | Speed (2–3 min vs. 20+ min manual); consistent across channels; firmware multi-dispense modes |
| Setting up 8 replicate dilutions manually, ad hoc adjustments | Single-channel | Flexibility to aspirate unique volumes; no tip alignment constraints |
| Occasional plate pooling or mixing in fixed-volume reactors | Manual 8-channel | Lower cost; no batteries; simple mechanics; adequate for low-frequency use |
| High-throughput PCR or ELISA with 100+ plates/week | Electronic 8 or 12 | Reduced RSI; improved repeatability; programmable speed; faster turnaround |
| Transferring from irregular tube racks to scattered microplate wells | Single-channel | Multichannel tips won’t align; forced to use single-channel anyway |
| Viscous sample (glycerol >20%, protein >5 mg/mL) with tight CV requirement | Electronic + reverse pipetting mode | Motor control of reverse technique minimizes CVs that are user-dependent in manual |
Cost-benefit summary:
- Electronic: Higher upfront cost (~$5,000–$15,000), lower operational strain, improved accuracy on repetitive tasks.
- Manual: Lower cost (~$500–$2,000), sufficient for sporadic use, acceptable accuracy if proper technique applied.
- Single-channel: Lowest cost, maximum flexibility, but slow and ergonomically demanding for high-volume work.
Key Modes and Functions (Practical)
Modern electronic multichannel pipettes offer programmable modes beyond basic aspiration–dispense. Understanding these modes unlocks speed and accuracy gains.
Multi-Dispense Mode
Aspirate once, dispense identical sub-volumes to multiple destinations without re-aspirating. Example: Aspirate 120 µL, then dispense 20 µL each to six wells in sequence. Saves time and reduces tip waste compared to six separate aspirations. Requires firmware configuration but is standard on most electronic units.
Mixing Mode
Pipette rapidly up and down within a single well to homogenize samples. Useful for resuspending beads or pooling reagents post-dispense. Programmable speed prevents bubble generation that would occur at very high pipetting speeds.
Serial Dilution Mode
Automatically decrement volume with each dispense step. Example: Aspirate 100 µL, dispense 50 µL to well 1, 40 µL to well 2, 30 µL to well 3, etc. Creates linear or logarithmic dilution series in seconds. Reduces calculation errors and accelerates assay setup.
Speed Control and Dwell Time
- Aspiration speed: Slow (50–100 µL/s) for viscous fluids and bubble-prone samples; fast (300–500 µL/s) for aqueous buffers.
- Dwell time: Programmable pause (0.1–2 s) at aspiration completion allows tip re-equilibration before withdrawal, reducing splashing and air bubbles.
- Dispense speed: Often faster than aspiration (300–800 µL/s) to save time.
When Reverse Pipetting Helps
Forward pipetting (aspire to first stop, dispense to first stop) is standard and ideal for aqueous solutions. Reverse pipetting-aspirating excess volume, dispensing the target amount, then discarding the remainder-reduces errors when pipetting viscous, foaming, or volatile liquids. Electronic pipettes with programmable reverse modes execute this consistently; manual reverse pipetting is more error-prone and user-dependent.
Pipette Tips & Compatibility (Critical for Multichannel)
Tip quality, fit, and material are often the limiting factors in multichannel accuracy-often more critical than the pipette itself.
Tip Fit and Sealing Across All Channels
Multichannel pipettes require simultaneous, uniform tip seating on all eight or twelve channels. Uneven pressure during tip loading creates a subtle slope: the outer tips seat differently than center tips, causing reproducible liquid-level differences (outer tips aspirate less). Always:
- Apply even, vertical pressure when loading tips-avoid rolling or angling.
- Use tips designed for the specific pipette model; universal tips may not achieve uniform seal.
- Visually inspect liquid levels in all tips after aspiration before dispensing; if uneven, re-seat tips and repeat aspiration once or twice.
Filtered vs Non-Filtered Tips
- Filtered (aerosol barrier) tips: Include a hydrophobic barrier to prevent aerosol contamination from splashing back into the pipette. Recommended for biosafety work, qPCR (to prevent cross-sample contamination), and ELISA washouts. Slight pressure difference; requires compatible pipette head.
- Non-filtered tips: Standard polypropylene; lower cost; acceptable for simple reagent transfers or non-critical workflows.
Low-Retention Tips
Viscous samples (glycerol, DMSO, protein solutions) adhere to standard tip walls, leaving measurable residue. Low-retention tips feature hydrophobic surface treatment to minimize stiction. Combined with reverse pipetting, they dramatically improve CV on challenging liquids.
Racked vs Bulk Tips (for speed and alignment)
- Racked tips (pre-loaded in trays): Aligned, ready to load; optimizes speed; ensures consistent positioning.
- Bulk tips (loose in containers): Cheaper; requires manual loading; more prone to misalignment if not inserted carefully.
Tip Seating & Seal Check Checklist
Before aspirating:
- ☐ Load tips with even pressure; no outer tips riding higher than center ones.
- ☐ Hold pipette vertically above tip box; ensure all tips make contact simultaneously.
- ☐ Tap tips once firmly into the pipette cone; audible click confirms seating.
- ☐ Place all tips into liquid; if any tips spray or drip, remove and reload.
- ☐ Aspirate a small volume; visually confirm liquid levels are identical across all tips.
Accuracy, Precision & Calibration (ISO 8655)
Understanding Accuracy vs Precision
- Accuracy (trueness): How close the delivered volume is to the nominal (programmed) volume. Example: You program 100 µL; the pipette delivers 101 µL. Error = +1%.
- Precision (repeatability): How consistent successive deliveries are. Example: Five 100 µL deliveries yield 99.8, 100.1, 100.3, 99.9, 100.4 µL. Standard deviation ≈ 0.2 µL; CV ≈ 0.2%.
ISO 8655:2022 specifies maximum permissible errors (accuracy) and repeatability (precision) thresholds for piston-operated volumetric apparatus including multichannel pipettes. Key updates from the 2002 standard:
- Pipette and tips treated as a system: Tips must be included in calibration; bare pipette performance is not sufficient.
- Stricter balance requirements: Six-place balances (microgram precision) for volumes <20 µL; five-place for 20–199 µL.
- Environmental controls: Temperature (±2°C), humidity (45–75% RH), and air pressure must be documented during calibration.
- Maximum tolerances vary by volume: Smaller volumes tolerate larger percent errors; larger volumes tighter absolute errors.
Calibration Frequency Guidance
- Heavy use (daily): Annually or every 6 months if handling viscous or problematic samples.
- Moderate use: Annually or as part of ISO 17025 accreditation protocols.
- After repair or tip change: If internal seals replaced or tip design changed, recalibrate.
Across-Channel Consistency Check (Simple in-house routine)
- Aspirate 50 µL of distilled water into all 8 channels.
- Dispense into 8 separate pre-weighed tubes.
- Weigh each tube; calculate mass of water (assume 1 mg ≈ 1 µL at 20°C).
- Calculate mean and standard deviation across 8 channels.
- Acceptable: All eight values within ±2% of 50 µL (i.e., 49–51 µL); CV <1%.
- Flag for service: If any channel deviates >2% or CV >1%, likely tip sealing or internal leakage; contact service.
Best Practices for Plate Pipetting (Step-by-Step)
Setup Steps
- Arrange plate(s) in front of you; ensure wells are not tilted (use plate holders if needed).
- Load pipette tips (8 or 12 channels depending on plate format).
- Fill reagent reservoir with sufficient volume; account for tip immersion (±20% extra).
- Power on pipette; set volume, speed (start: 100 µL/s aspiration, 200 µL/s dispense), and dwell time (start: 0.5 s).
Forward Pipetting Steps (Standard for aqueous solutions)
- Position pipette vertically above tip box; apply gentle, even pressure downward.
- With all tips in the reagent reservoir, hold pipette at 90° angle without touching walls.
- Press plunger to first stop smoothly; release slowly, allowing liquid to rise to aspirate mark.
- Wait one second (dwell) for equilibration.
- Withdraw tips vertically from reservoir.
- Move to microplate; position tips at an angle ~45° to well walls (not straight down).
- Immerse tips 2–3 mm into target wells; press plunger to first stop to dispense.
- Hold for dwell time (~0.3 s), then withdraw tips.
- Discard tips into waste; repeat for next column.
Reverse Pipetting Steps (For viscous, foaming, or volatile samples)
- Set volume to target volume + excess (e.g., for 100 µL delivery, set 120 µL).
- Press plunger to second stop (full blowout), then insert tips into liquid.
- Slowly release plunger to full extension, aspirating 120 µL.
- Withdraw tips; move to target well.
- Press plunger to first stop only to dispense 100 µL; stop and hold.
- Discard the remaining 20 µL by pressing to second stop into a waste container.
- Do not blow out into the target well (excess liquid wastes material and may compromise assay).
Consistent Immersion Depth and Angle
- Depth: Always aim for 2–3 mm; deeper immersion increases backpressure and risk of bubble carryover.
- Angle: At aspirate, hold ~90° (vertical) for even pressure. At dispense, tilt ~45° to well wall to reduce splashing and aerosol generation.
- Speed adjustment by liquid: Slow for glycerol (100 µL/s), fast for water (400 µL/s), moderate for buffers (200 µL/s).
Avoiding Bubbles and Splashing
- Pre-wet tips (one aspirate–dispense cycle) with sample if using low-surface-tension liquids.
- Tap tip box firmly once to seat all tips before first aspirate.
- Never aspirate or dispense at maximum speed into a full well; risk of aerosol carryover.
- If bubbles are visible in tips after aspiration, gently pipette up–down twice within the reservoir to dislodge them before moving to the plate.
Troubleshooting (Must Include)
| Symptom | Likely Cause | Quick Fix | When to Service |
|---|---|---|---|
| Uneven volumes across channels | Tips not seated evenly; bent outer tips; internal channel leak | Reload tips with extra-firm even pressure; inspect for bent tips; check for liquid pooling inside pipette head | If persists after reload; service internal seals |
| Bubbles in tips during aspiration | Aspirating too fast; air gap in reservoir; tips not reaching liquid | Reduce speed 50%; ensure reservoir liquid level is adequate; reposition tips deeper into liquid | N/A (user technique) |
| Dripping from tips at rest | Poor tip fit; worn internal O-rings; over-filled tips | Remove and re-seat tips; check O-ring condition; avoid over-aspiration | If O-rings visibly damaged or dripping persists |
| Tip sealing issues (spray during use) | Bent or defective tips; pipette head misalignment | Inspect each tip; discard any that are bent; remove and reinstall tip-loading cone | Misalignment; contact manufacturer |
| Mode errors (multi-dispense not working as programmed) | Wrong mode selected; protocol not saved; low battery | Verify mode in display; re-enter and save protocol; check battery level | No; user error likely |
| Inconsistent battery performance (intermittent operation) | Aging battery; poor contact | Charge fully; clean battery contacts with dry cloth | Replace battery after 2–3 years typical use |
Ergonomics and Fatigue (Why People Upgrade)
Almost 90% of laboratory workers who pipette continuously for >1 hour per day report hand, wrist, elbow, or shoulder pain. Repetitive strain injuries (RSI) are caused by three factors: force, repetition, and poor posture.
Reduced Thumb Strain with Electronic Multichannel Pipettes
- Manual pipettes require 2–5 N thumb force per stroke. Across 100+ strokes per day, cumulative tendon loading in the thumb flexor causes tenosynovitis (inflammation of the tendon sheath).
- Electronic pipettes: Button press requires <1 N; motor does the work. Over a full day, this reduces thumb loading by ~70%.
- Multichannel vs single-channel: Filling a 96-well plate with an 8-channel pipette = 12 button presses. Single-channel = 96 button presses. Electronic 8-channel eliminates 84 strain cycles daily.
Grip and Posture Tips
- Hold the pipette with fingers, not palm; avoid death-grip tension. Excessive grip force translates to forearm muscle fatigue and wrist tension.
- Keep wrist straight (not bent forward or back) while pipetting; wrist bending is a major RSI risk factor.
- Elbows close to body (not flared out); minimizes shoulder elevation.
- Maintain neutral neck posture: Avoid tilting head to look at microplates; bring plate up to eye level using plate stands.
- Take 1–2 minute breaks every 20 minutes if pipetting continuously; stretch hands and wrists.
Work Habits to Reduce RSI
- Switch to multichannel (manual or electronic) for any plate-based workflow; reduces single-channel repetitions drastically.
- Use electronic multichannel if pipetting >2 hrs/day; the motor assistance is clinically significant in reducing strain.
- Use adjustable-tip-spacing multichannel for mixed workflows (tubes + plates); maintains efficiency while reducing forcing into rigid formats.
- Rotate left and right hand use if using single-channel, even if you’re right-handed; distribute fatigue.
FAQ
Q1: Are electronic multichannel pipettes more accurate than manual?
Yes, in repeatability (precision). Electronic pipettes deliver the same piston velocity on every stroke, achieving CVs of 0.5–1% across channels; manual multichannel CV is typically 1–2% due to variable thumb speed. However, both depend critically on tip sealing and immersion depth. Poor technique negates electronic precision.
Q2: What’s the best channel count for 96-well plates?
8-channel is standard and fastest for full 96-well fills (12 operations). However, if your workflow mixes 96-well and 384-well plate work, and you can afford two pipettes, 8-channel for 96-well and 12-channel for 384-well reduces user error from repeated tip reloading. If single pipette only: choose 8-channel for versatility.
Q3: Why do some channels aspirate differently?
Most common cause: uneven tip seating during loading. Outer tips may seat slightly higher or lower than center tips, creating tilted loading angle. Result: outer tips experience different backpressure and aspirate less volume. Fix: Reload tips with firmer, more deliberate even pressure across all eight channels. Second cause: tip manufacturing defects or bending; inspect visually and discard bent tips.
Q4: Are electronic multichannel pipettes worth the cost?
Depends on volume. If your lab pipettes <100 plates/week, a manual 8-channel suffices and saves $10,000+. If >500 plates/week or RSI is a concern: electronic multichannel pays for itself in reduced ergonomic liability and faster output in 2–3 years. Additionally, programmable modes (multi-dispense, serial dilution, reverse pipetting) reduce protocol execution time by 30–50% on complex assays.
Q5: How often should I calibrate my electronic multichannel pipette?
Annually for normal use (ISO 8655 compliance). If handling viscous samples (>20% glycerol, protein solutions), calibrate every 6 months. After internal service or tip type change, recalibrate before resuming work. Many labs do in-house accuracy checks quarterly using the gravimetric method (weigh 10× 50 µL dispensed into tubes) to extend calibration intervals.
Key Takeaways
- Electronic multichannel pipettes eliminate thumb strain while maintaining sub-1% CV across channels, ideal for >500 plates/week or when RSI is a concern.
- Tip seating and fit are more critical to accuracy than the pipette itself; invest in high-quality, certified tips and load with firm, even pressure.
- Channel count choice (8 vs 12) depends on plate format: 8 for 96-well, 12 for 384-well; choose once and stick unless workflow demands both.
- Reverse pipetting mode reduces errors on viscous/volatile samples by ~2–3%, but requires compatible filtered tips with sufficient dead space.
- Programmable speed control and dwell time are underused; customizing for sample viscosity cuts CVs by 30–50% compared to fixed defaults.
- Calibration is mandatory annually under ISO 8655:2022; stricter balance requirements (6-place for <20 µL) and environmental controls ensure traceability.
- Multi-dispense and serial dilution modes accelerate plate setup by 30–50% and reduce calculation errors; learn these workflows to justify the instrument cost.
- Manual multichannel is sufficient for sporadic, low-throughput work; electronic pays off at >300 plates/week in both time savings and ergonomic benefit.
- Ergonomic upgrades (low-force buttons, electronic tip ejection, multi-finger triggers) reduce daily RSI risk by ~70% compared to traditional designs.
- Environmental controls during use (room temperature ±2°C, humidity 45–75%) maintain calibration accuracy; seasonal recalibration may be needed in uncontrolled labs.