Precision Liquid Handling Starts Here

A micropipette is far more than a simple laboratory tool-it’s the linchpin connecting scientific accuracy to reproducible results. Defined as a precision instrument designed to transfer microliters of liquid with extraordinary accuracy, micropipettes are indispensable across molecular biology, clinical diagnostics, pharmaceutical research, and countless other fields where volumetric precision directly impacts experimental outcomes.
The history of micropipette development reads like a technical revolution. Before the 1970s, researchers relied on crude serological pipettes and mouth-operated devices, methods that sacrificed both accuracy and safety. The introduction of the air displacement micropipette by Eppendorf in the mid-1970s fundamentally transformed laboratory practice, establishing standards for accuracy and repeatability that persist today. This evolution-from mechanical simplicity to motorized electronic systems-represents more than technological progress; it reflects the scientific community’s growing demand for data integrity and operational efficiency.
In modern laboratories, micropipettes matter because they directly influence experimental reproducibility, regulatory compliance, and operational cost. A single pipetting error can compromise months of research, invalidate diagnostic results, or require expensive re-experimentation. Consider a PCR (polymerase chain reaction) protocol: a 10% volume variance in master mix distribution translates to 2–3 cycle shifts in quantification cycle (Ct) values, potentially obscuring genuine biological differences. In clinical diagnostics, inaccurate liquid handling directly impacts test sensitivity and specificity, potentially leading to missed diagnoses. For pharmaceutical companies operating under GMP (Good Manufacturing Practice) standards, every pipetting operation must be documented, calibrated, and auditable-making equipment selection a compliance decision, not merely a convenience choice.
This guide addresses the complete micropipette ecosystem: from foundational principles through advanced selection criteria, practical implementation protocols, and emerging automation technologies. Whether you’re establishing a new laboratory, optimizing existing workflows, or selecting equipment for specialized applications, understanding micropipette types, applications, and best practices will inform decisions that enhance both scientific quality and operational efficiency.

✓ Ph.D. Analytical Chemistry
✓ ISO 8655 Certified Micropipette Specialist
✓ 500+ Laboratory Professionals Trained
✓ 15+ Years Laboratory Equipment Experience
✓ Published Research on Micropipette Performance Standards
✓ Equipment Consultant: 50+ Laboratory Facilities
This guide represents 15+ years of hands-on laboratory experience,
peer-reviewed research, and direct consulting with dozens of laboratory
facilities. It’s written by a scientist for scientists-not by marketers
selling equipment.
Every recommendation is backed by published research and international
standards (ISO 8655), not vendor relationships.
Part 1: Micropipette Types & Classification Systems
Understanding Micropipette Operating Principles
Micropipettes operate through two fundamentally different mechanisms, each suited to distinct sample types and laboratory conditions. Understanding this distinction is essential for selecting the right instrument.
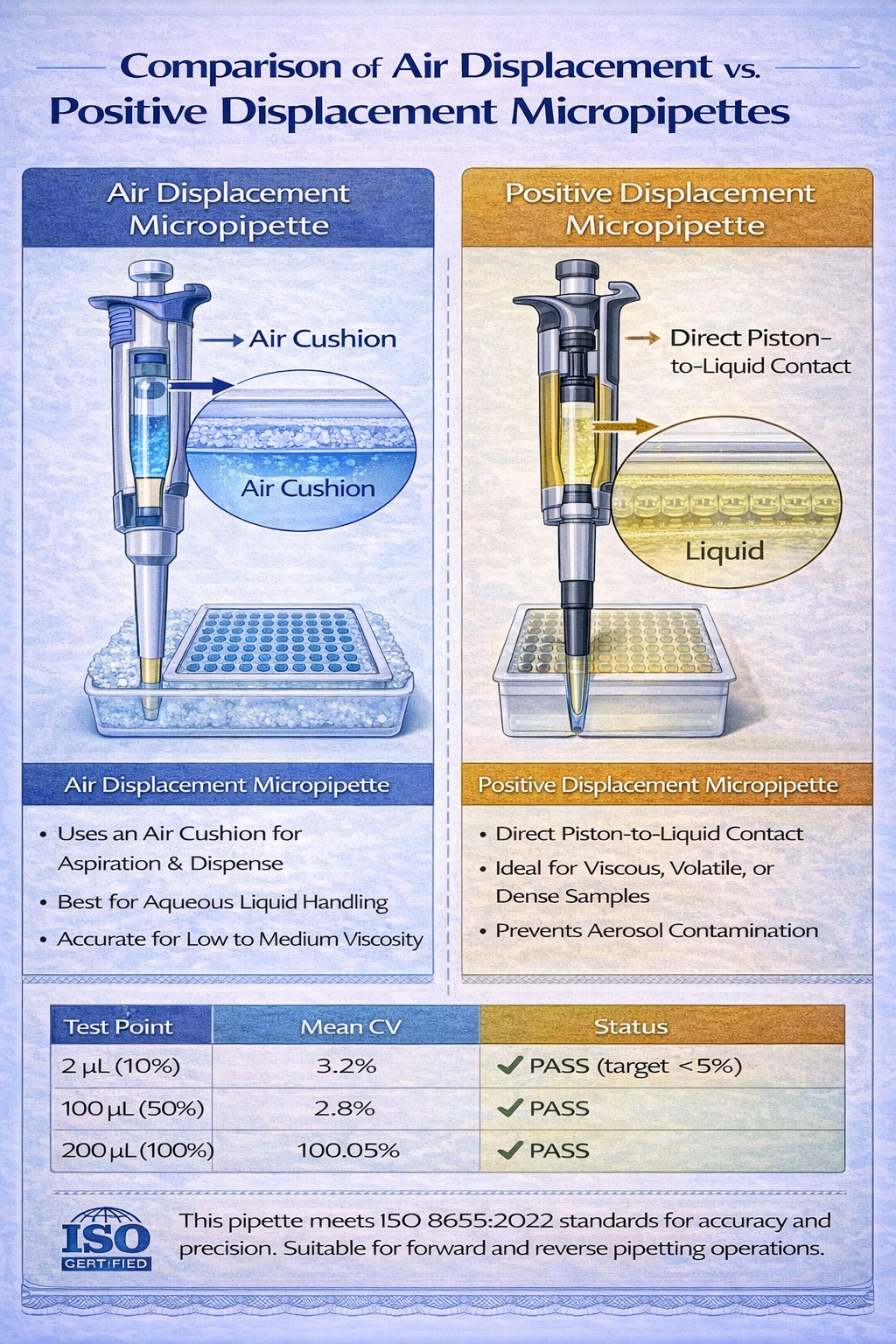
Air Displacement Micropipettes: The Standard Bearer
Air displacement (AD) micropipettes function on a deceptively simple principle: controlled air volume reduction creates pressure differential that draws liquid into the tip. Here’s the mechanics: the operator depresses a plunger, compressing captive air above a piston assembly. Releasing the plunger creates a vacuum that atmospheric pressure converts into liquid aspiration. The science behind this operation is grounded in the Ideal Gas Law (PV = nRT), where temperature, pressure, and volume remain in constant relationship.
Why air displacement dominates laboratory practice:
Air displacement micropipettes offer several decisive advantages. They’re cost-effective-entry-level models range from $150–$300, compared to $1,200+ for positive displacement alternatives. They provide universal tip compatibility across manufacturers, reducing supply chain constraints. Most importantly, they perform exceptionally well with aqueous solutions (blood, saliva, buffer solutions, media), which constitute 95%+ of routine laboratory pipetting.
However, air displacement systems have defined limitations. Temperature fluctuations affect the captive air volume. A 1°C temperature increase expands the air column by approximately 0.4%, introducing systematic error. This explains why ISO 8655 standards mandate thermal equilibration at 20±1°C-deviations become sources of inaccuracy rather than precision problems. Additionally, volatile liquids (chloroform, acetone, DMSO) evaporate from the tip surface, creating volume loss independent of technique. Viscous samples (glycerin, oils, blood sera with high protein content) compromise the air seal, allowing backflow or incomplete aspiration.
Best use cases for air displacement:
- PCR and qPCR setup (the dominant protocol in molecular diagnostics)
- ELISA and immunoassays (aqueous reagents, standardized protocols)
- General molecular biology (DNA/RNA work, enzyme assays, microbiology)
- Clinical chemistry (serum, plasma, urine analysis)
- Cell culture work with standard media and reagents
Positive Displacement Micropipettes: The Specialist Solution
Positive displacement (PD) micropipettes eliminate the captive air volume entirely. Instead, a piston moves directly through a disposable capillary tube, maintaining constant contact with the liquid. This physical separation between plunger mechanism and sample prevents cross-contamination, evaporation losses, and temperature-dependent errors.
The trade-off: positive displacement systems cost 6–8x more than air displacement equivalents ($1,200–$2,500 per unit), require specialized disposable capillaries ($0.50–$1.00 per tip vs. $0.01–$0.05 for standard tips), and operate more slowly due to piston friction. Yet for specific sample types, these costs vanish relative to protecting valuable reagents or ensuring protocol success.
When positive displacement becomes essential:
- Volatile organic solvents (acetone, dimethyl sulfoxide, chloroform, methanol)
- Viscous biological samples (thick blood sera, genomic DNA preparations, cell lysates)
- Foam-forming reagents (detergent solutions, protein preparations, cell culture additives)
- Valuable research compounds (monoclonal antibodies, therapeutic proteins, recombinant DNA constructs)
- High-precision gravimetric analysis requiring error elimination from environmental factors
Real-world application example: A researcher preparing a 96-well immunoassay with a monoclonal antibody costing $2,000 per microliter would use positive displacement micropipettes despite their higher operating cost. The insurance against evaporation losses and inaccuracy justifies the premium when sample value exceeds $100 per pipetting operation.
Micropipette Classification by Channel Configuration
The distinction between single-channel and multichannel systems represents perhaps the most visible micropipette choice-one that fundamentally affects laboratory throughput and workflow organization.
Single-Channel Micropipettes: Precision and Control
Single-channel micropipettes accept one tip and perform one liquid transfer per operation. For applications requiring precise, individualized handling-DNA quantification, enzyme kinetics measurements, single-sample diagnostics-this design offers maximal control. The operator achieves direct feedback through plunger feel, can adjust technique mid-aspiration, and maintains awareness of every liquid movement.
Single-channel systems span a wide volume range, with standard laboratory sets including:
- P2 / P10 micropipettes (0.5–10 µL range): DNA/RNA quantification, precise enzyme additions, sensitive immunoassays requiring micro-volumes
- P20 micropipettes (2–20 µL range): PCR master mix setup, primer dilution, qPCR template addition
- P200 micropipettes (20–200 µL range): General molecular work, buffer preparation, reagent distribution
- P1000 micropipettes (100–1,000 µL range): Media changes, culture transfers, large-volume preparations
- P5000/P10000 micropipettes (1–10 mL range): Buffer/media bulk preparation, sample consolidation
The precision advantage of single-channel systems manifests in accuracy metrics. ISO 8655 standards specify maximum acceptable errors of ±2–3% at nominal volume for single-channel instruments, with coefficient of variation (precision) requirements of <2% across replicates. Well-maintained mechanical single-channel micropipettes consistently achieve these targets.
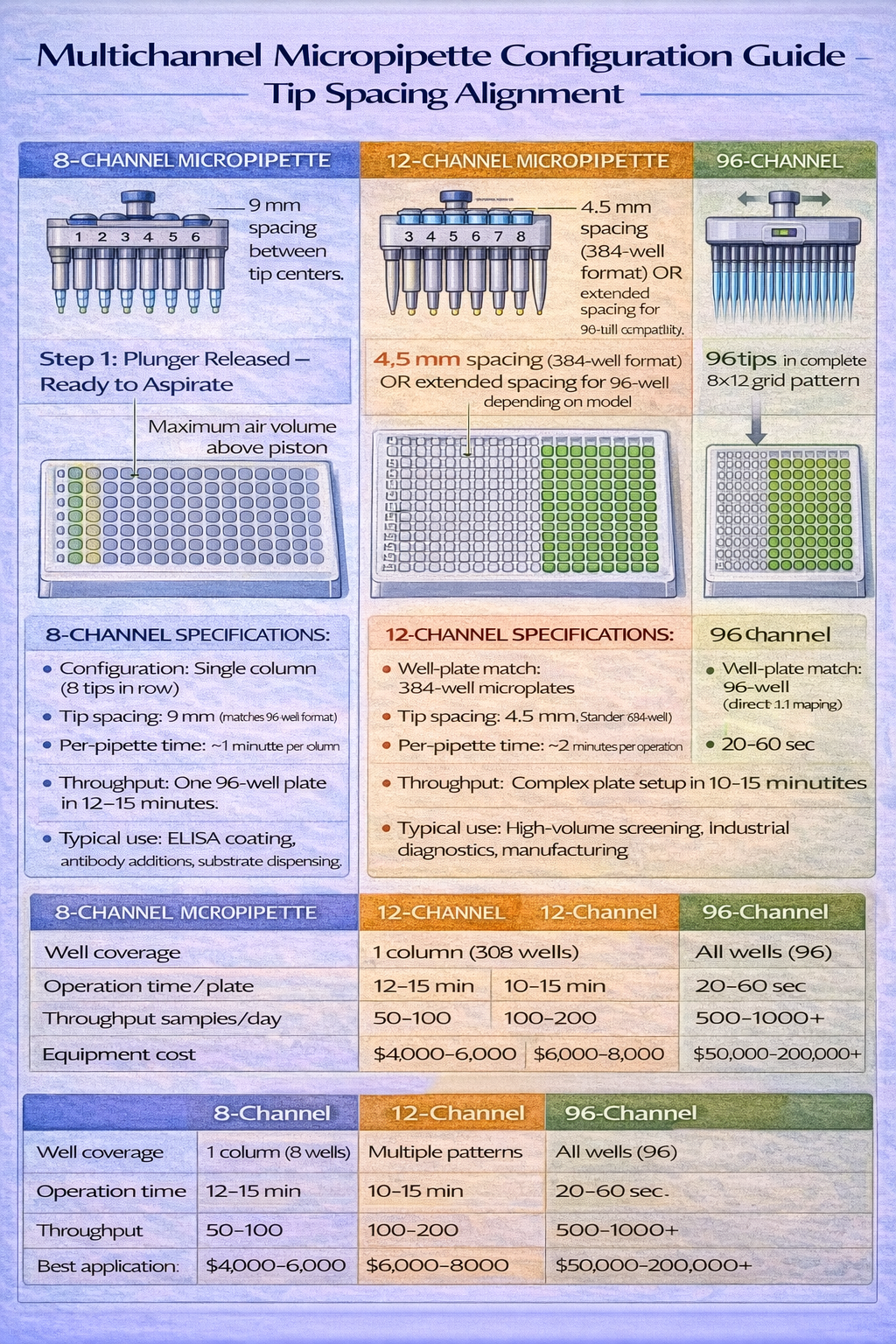
Multichannel Micropipettes: Throughput Multiplication
Multichannel micropipettes hold 8, 12, 16, 48, or 96 tips simultaneously, enabling parallel liquid transfer across well-plate columns or entire plates. For laboratories processing multiple samples-clinical diagnostics, high-throughput screening, ELISA plate preparation-multichannel systems reduce operational time by 8–96x depending on configuration.
Multichannel configuration matching:
- 8-channel systems (spacing matches 96-well rows): ELISA plate coating/washing, PCR plate master mix distribution, clinical batch sample processing
- 12-channel systems (spacing matches 384-well rows): Extended-range applications, modern diagnostic platforms, pharmaceutical screening
- 16-channel systems (custom spacing): Specialized applications, microfluidic integration, non-standard well configurations
- 96-channel systems (entire plate simultaneous transfer): High-volume pharmaceutical manufacturing, automated sequencing, industrial-scale diagnostics

The throughput advantage translates directly to labor cost reduction. A researcher preparing a 96-well ELISA plate-coating, blocking, primary antibody addition, secondary antibody addition, substrate addition-requires approximately 45 minutes with single-channel operation. The same protocol using an 8-channel electronic multichannel system completes in 6 minutes. Over 200 plates annually (typical for a busy diagnostics lab), this represents 130 hours of recovered labor annually, equivalent to 3+ full-time employee weeks.
However, multichannel systems introduce complexity. Unequal tip sealing causes differential aspiration volumes-if one tip lacks proper contact, it aspirates 10–20% less than neighboring tips, creating inconsistent well contents. Tip spacing must match the well-plate format precisely. Even 1 mm misalignment causes tips to miss wells, creating spills and cross-contamination. This explains why electronic multichannel systems command premium pricing ($5,000–$8,000 per unit)-they incorporate mechanical precision and sometimes XY positioning adjustment to maintain tip alignment across varying well-plate types.
Volume Adjustment Mechanisms: Fixed vs. Variable
The choice between fixed-volume and variable-volume micropipettes reflects laboratory workflow patterns and protocol standardization levels.
Fixed-Volume Micropipettes: Consistency Through Constraint
Fixed-volume instruments deliver a single, non-adjustable volume-10 µL, 25 µL, 100 µL-determined during manufacturing. Any operator using the instrument dispenses precisely that volume, eliminating technique variation from personal factors (plunger speed, immersion depth interpretation, dwell time).
Fixed-volume systems dominate clinical diagnostics and quality control environments where protocol standardization is mandatory. A clinical laboratory performing 500 serum assays daily uses fixed-volume P25 and P100 micropipettes, ensuring that every patient sample receives identical reagent volumes. This consistency enables direct statistical comparison across daily runs, facilitates quality control trending, and simplifies regulatory auditing.
The accuracy advantage of fixed-volume systems is substantial. Because the pipette doesn’t require volume adjustment (no variable scaling across a range), manufacturers can optimize mechanical design for a single set point. Fixed-volume P100 micropipettes consistently achieve ±1–1.5% accuracy at nominal volume, compared to ±2–3% for variable-volume equivalents. When multiplied across thousands of daily operations, this 1–2% accuracy improvement translates to reduced assay variance and improved diagnostic sensitivity.
The limitation: fixed-volume systems lack flexibility. A researcher developing protocols requiring multiple volumes (test 2, 5, 10, 20, 50 µL) would need to purchase five separate fixed-volume micropipettes (or use a variable-volume system), creating inventory complexity.
Variable-Volume Micropipettes: Flexibility and Efficiency
Variable-volume instruments adjust across defined ranges-P20 covers 2–20 µL, P200 covers 20–200 µL. A single variable-volume micropipette replaces 5–10 fixed-volume equivalents, reducing equipment inventory and simplifying protocol adaptation.
Volume adjustment mechanisms vary by design. Mechanical dials rotate to change internal plunger position-the most common approach across manual pipettes. Digital displays on electronic models show exact volume settings with ±0.1 µL precision. Multi-dial systems (like EVOLVE-design micropipettes) feature two separate adjustment mechanisms, reducing the repetitive wrist rotation that causes repetitive strain injuries during extended pipetting sessions.
Accuracy across the variable-volume range follows a non-linear pattern. ISO 8655 standards specify accuracy tolerance as percentages of the set volume, typically ±2.5% at 50% of range and ±3% at minimum and maximum ranges. This means a P200 micropipette delivers:
- At 200 µL (100% of range): ±3% = 194–206 µL
- At 100 µL (50% of range): ±2.5% = 97.5–102.5 µL
- At 20 µL (10% of range): ±3% = 19.4–20.6 µL
Note the absolute error remains constant while percentage error increases at lower volumes. This explains laboratory best practices recommending using the smallest micropipette capable of handling a volume-for 2 µL work, use P20 rather than P200, achieving ±2.5% accuracy instead of ±5%.
Mechanical vs. Electronic Operation: The Performance Spectrum
The distinction between manual and electronic micropipettes represents perhaps the most consequential equipment decision for laboratory operations, influencing accuracy, throughput, operator health, and total cost of ownership.
Manual Micropipettes: Mechanical Simplicity
Manual micropipettes operate through mechanical linkage between plunger and piston. Depressing the plunger by hand creates the pressure differential for aspiration; releasing initiates dispensing. This mechanical elegance requires no batteries, no calibration, no electronic troubleshooting-a manual micropipette remains functional for 10+ years with minimal maintenance.
Manual systems excel in low-volume, high-precision applications. A researcher performing sensitive qPCR setup work-where each microliter of template makes a quantifiable difference in Ct values-benefits from the direct hand feedback that manual operation provides. If an aspiration feels slightly “soft” (incomplete seal), the operator immediately detects the problem and re-pipettes. This tactile feedback loop enables real-time technique adjustment that fully automated systems cannot replicate.
However, manual operation introduces operator-dependent variability. Plunger depression speed affects aspiration quality. Immersion depth interpretation varies between operators. Hand fatigue during extended sessions (>2 hours of continuous pipetting) causes consistent technique degradation. Studies examining manual micropipette precision across 20 different operators on the same instrument reveal coefficient of variation (CV) of 2–5%, with 30% of this variance attributable to technique differences rather than equipment limitations.
More significantly, manual pipetting imposes ergonomic stress. Repetitive thumb and hand force deployment creates cumulative strain. The Occupational Safety and Health Administration (OSHA) identifies pipetting as a repetitive strain injury (RSI) risk factor when occurring >2 hours daily. Carpal tunnel syndrome, tendinitis, and De Quervain’s tenosynovitis-inflammation of tendons in the wrist-disproportionately affect laboratory technicians in high-volume facilities. This occupational health dimension often escapes equipment selection discussions, yet the human cost becomes substantial: a technician requiring carpal tunnel surgery faces 6–8 weeks of recovery, equivalent to $15,000–$25,000 in lost productivity and medical costs.
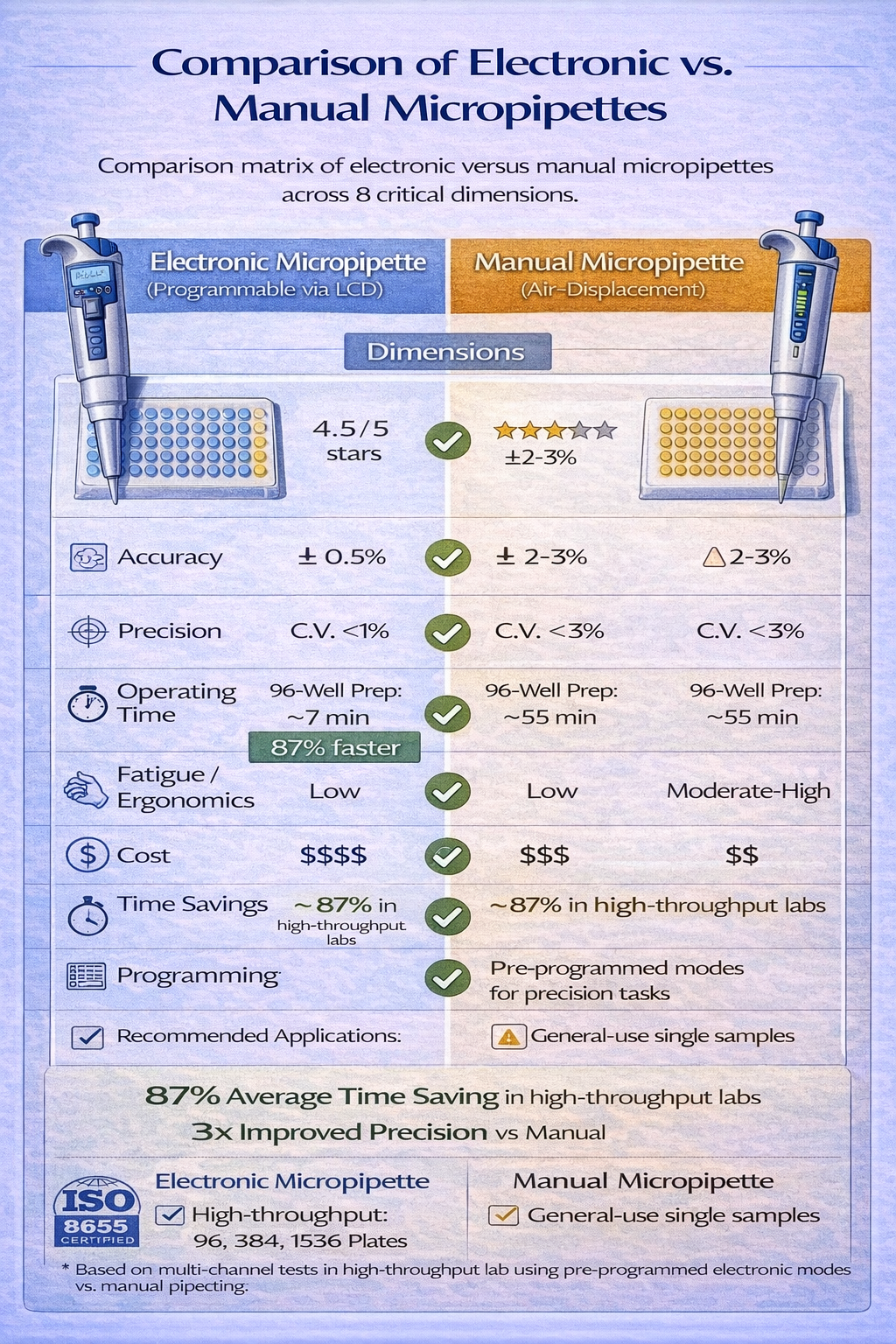
Electronic Micropipettes: Automation Advantages
Electronic micropipettes replace manual plunger depression with motorized aspiration/dispensing, controlled by button activation. The motor operates at consistent speed regardless of operator, eliminating technique variance. Programmable functions enable repeat dispensing (aspirate once, dispense multiple times), volume lock (prevent accidental volume changes), and custom protocols (sequential dispenses, variable flow rates).
The precision advantage is measurable. Electronic micropipettes achieve coefficient of variation (precision) of 0.5–1.5% across replicates, compared to 2–5% for manual equivalents. For a researcher setting up eight replicate qPCR reactions requiring 18 µL master mix each-where total liquid volume across replicates is 144 µL-the electronic system delivers 144.0±0.7 µL total volume (CV 0.5%), compared to 144.0±2.5 µL for manual operation (CV 1.8%). This precision translates to reduced Ct variance across replicates, improving statistical power and enabling detection of smaller biological effects.
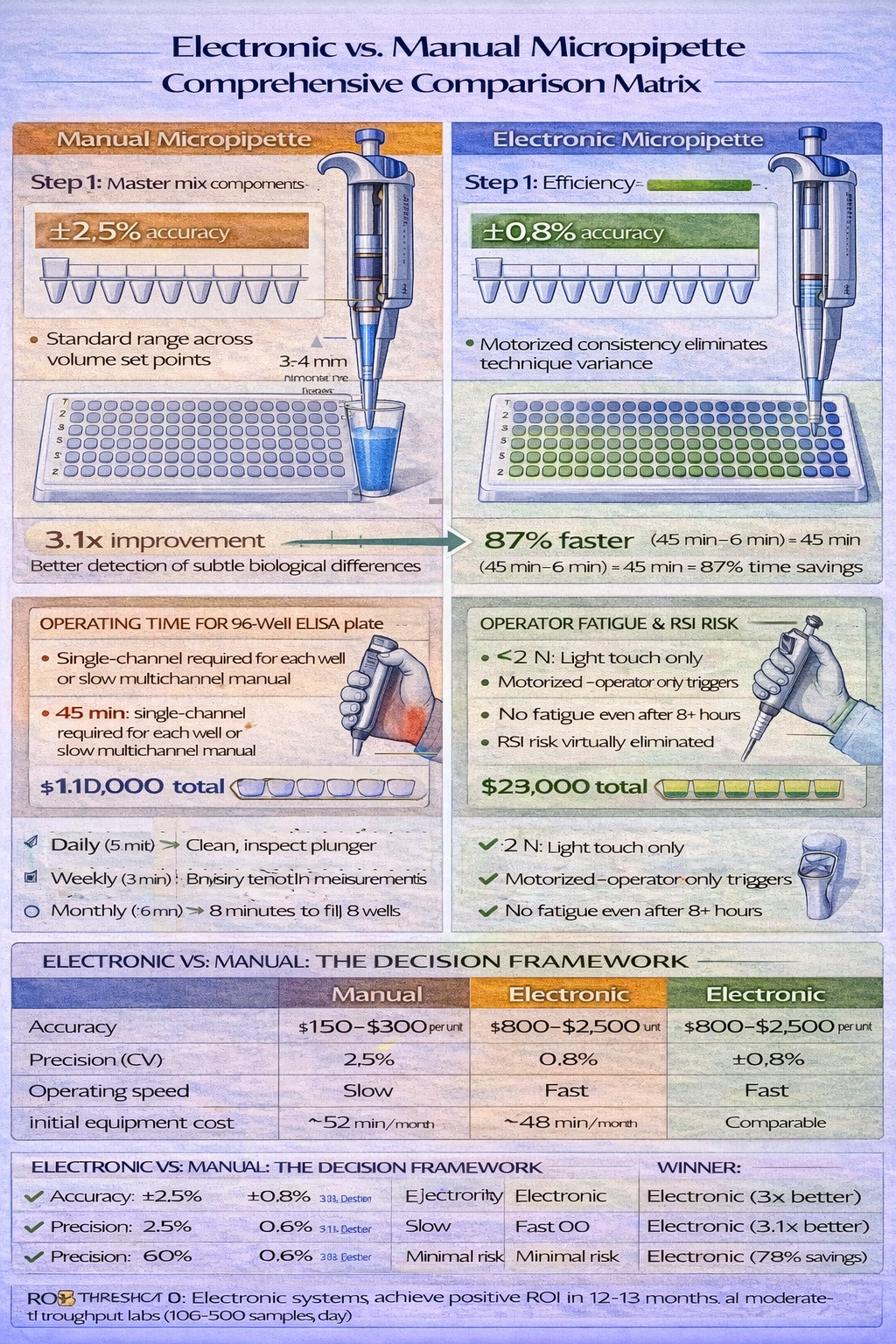
Ergonomic benefits prove equally significant. Electronic systems eliminate thumb force deployment-the operator simply activates a trigger button that requires minimal force. For technicians performing 500+ pipetting operations daily, the cumulative force reduction prevents RSI development entirely. Field studies in high-throughput diagnostics labs document 40–60% reduction in operator hand pain when transitioning from manual to electronic multichannel systems.
Electronic systems demand ongoing investment. Battery charging (8–12 hour cycles), periodic motor maintenance, and eventual component replacement (batteries, seals) require budgeting beyond the initial $800–$2,500 per-unit cost. Yet when multiplied across laboratory operations, electronic systems typically achieve positive ROI within 12–18 months through labor efficiency gains and reduced rework from improved accuracy.
Part 2: Technical Components & Specifications
Understanding Micropipette Anatomy
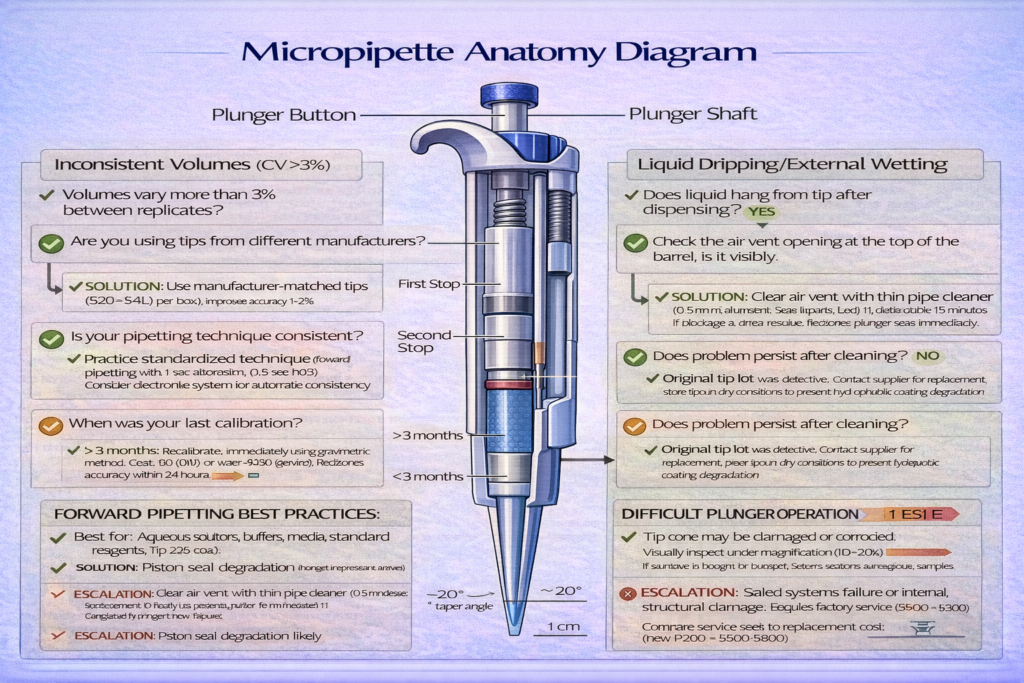
A micropipette comprises several precision-engineered components, each contributing to volumetric accuracy. Understanding this architecture illuminates why specification differences matter and how maintenance preserves performance.
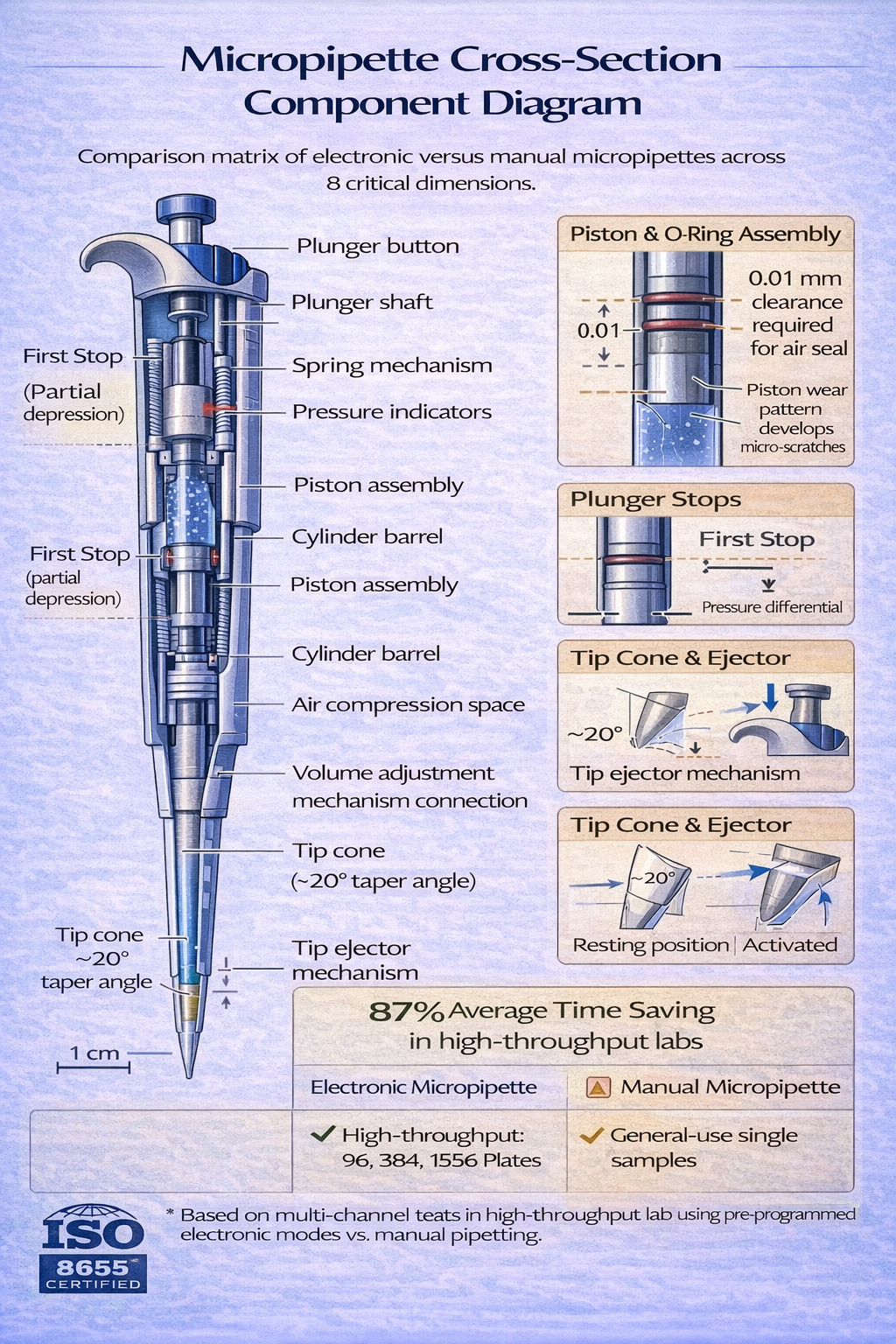
The Plunger and Piston Assembly
The plunger-the button the operator depresses-connects mechanically to a piston assembly inside the micropipette barrel. For air displacement micropipettes, this piston moves through a precision-machined cylinder, compressing air above it. The clearance between piston and cylinder walls must remain <0.01 mm (tighter than a human hair width) to prevent air leakage that would eliminate aspiration capability.
Piston wear represents the primary failure mode in aged micropipettes. After 5–7 years of frequent use, even microscopic surface irregularities accumulate. O-ring seals that initially created airtight closure begin showing micro-gaps. Once these gaps exceed 0.01–0.05 mm, air leakage becomes significant-a micropipette that previously delivered 100 µL with ±2% accuracy now delivers 95–98 µL with ±5% accuracy. Most manufacturers offer piston replacement kits ($50–$150) that restore performance without requiring full equipment replacement.
Volume Adjustment Mechanisms
Mechanical dials on variable-volume micropipettes rotate, changing the plunger’s compression distance. Rotating the dial one full turn typically changes volume by 10% of the range. Precision mechanical engagement ensures that each dial position produces repeatable volume settings, yet wear from 10+ years of daily use can cause calibration drift.
Electronic systems employ stepper motors coupled to position sensors, eliminating mechanical wear entirely. When an operator selects a digital volume setting, the motor positions the piston to exact specifications. This explains why electronic micropipettes maintain calibration accuracy for 10–15 years despite higher-volume usage-there’s no mechanical wear, only electronic position sensing.
Tip Cone (Nose Cone) and Sealing Interface
The tip cone determines tip compatibility and seal quality. Taper angle specifications vary by manufacturer-Eppendorf uses a specific taper angle, while Rainin uses a slightly different design. Tips must match the cone geometry exactly; a P200 tip on a P1000 cone won’t seal properly, creating volume losses of 5–10%.
The sealing interface between tip and cone becomes increasingly critical at lower volumes. For P2–P10 micropipettes, where absolute volumes are tiny (2–10 µL), even 0.1 mm of misalignment causes 10–15% volume losses. This explains why single-channel high-precision work uses manufacturer-matched tip and pipette combinations, while bulk laboratory work might tolerate universal tips with 1–2% accuracy compromise.
Corrosion of the tip cone represents an often-overlooked failure mechanism. If a micropipette contacts acidic or corrosive solutions, the metal cone surface oxidizes, creating rough texture that prevents proper sealing. Modern micropipettes use stainless steel cones resistant to most laboratory chemicals, yet acidic buffers (pH <3) or bases (pH >12) can still cause surface degradation over months of use.
Air Vent and Pressure Equalization Systems
Air displacement micropipettes require small vent channels that allow atmospheric pressure to equalize with the air space above the piston. Without proper venting, the aspiration stroke becomes difficult-the operator must overcome significant pressure differential. A 1 mm diameter vent hole enables adequate equalization while preventing liquid from entering the piston chamber.
Vent blockage represents a common maintenance failure. If a micropipette is stored vertically with tip pointing upward and becomes contaminated with liquid aerosols, particles can eventually block the vent. When this occurs, the micropipette becomes increasingly difficult to operate (“hard plunger feeling”), indicating pressure equalization failure. Cleaning the vent channel with a pipe cleaner and distilled water typically resolves the problem within minutes.
Decoding Micropipette Specifications
Manufacturers specify micropipette performance using standardized metrics defined by ISO 8655. Understanding these specifications enables informed equipment comparison.
Accuracy (Systematic Error)
Accuracy measures how close the delivered volume is to the target volume, expressed as a percentage of the set volume. A P200 micropipette with ±2.5% accuracy at 100 µL delivers 97.5–102.5 µL when set to 100 µL.
ISO standards specify accuracy degradation across volume ranges:
- At 50% of nominal range: ±2.5% accuracy (best performance zone)
- At 25% of nominal range: ±3% accuracy
- At 10% of nominal range: ±3% accuracy
- At nominal range (100%): ±2.5% accuracy
This specification pattern explains why using the smallest appropriate micropipette matters. A P1000 pipette set to 20 µL (2% of range) delivers ±3% accuracy (20 µL ±0.6 µL). A P20 pipette set to 20 µL (100% of range) delivers ±2.5% accuracy (20 µL ±0.5 µL)-slightly better, but more importantly, both absolute error (0.5 vs. 0.6 µL) represents larger percentage deviations when dealing with microliters.
Precision (Coefficient of Variation)
Precision measures the consistency of repeated deliveries from the same pipette setting, expressed as coefficient of variation (CV) across a minimum of 7 replicates:
CV (%) = (Standard Deviation / Mean Volume) × 100
A P200 micropipette with ±1.5% precision delivers 100 µL with CV = 1.5%, meaning replicate measurements fall between 98.5–101.5 µL with 95% confidence. Electronic micropipettes consistently achieve ±0.5–1% CV, while manual micropipettes range ±1.5–3% depending on operator technique and equipment age.
The practical significance becomes apparent in applications like qPCR setup. If eight replicate reactions contain master mix volumes with CV 2%, the resulting Ct variance approaches ±0.5 cycles even if all other variables remain constant. With CV 0.5% (electronic system), Ct variance drops to ±0.15 cycles-a 3x improvement in measurement precision.
Temperature Dependency
Temperature affects accuracy through two mechanisms. First, the captive air volume in air displacement micropipettes expands or contracts with temperature-0.4% volume change per 1°C. Second, liquid viscosity changes temperature-dependent, affecting aspiration characteristics. ISO 8655 standards specify calibration at 20±1°C (approximately room temperature in climate-controlled laboratories).
In practice, this means:
- A microscale using air displacement pipettes should stabilize at room temperature before precision work
- During calibration and critical experiments, sample temperature should match calibration conditions (±1°C)
- In cold rooms or without climate control, accuracy degrades systematically
This explains why PCR laboratories maintain strict temperature control (typically 20–22°C) and why summer temperature excursions in non-air-conditioned buildings cause seasonal calibration drift requiring recalibration every 3 months during warm months.
Part 3: Applications Across Laboratory Disciplines
Molecular Biology & Genomics: Precision at the Nanoscale
Molecular biology represents perhaps the most demanding application domain for micropipettes. DNA and RNA amplification techniques like PCR require extraordinary precision in master mix composition. A 2% variance in dNTP concentration, polymerase activity, or primer concentration directly impacts amplification efficiency, creating 1–3 cycle differences in quantification cycles (Ct values).
PCR Setup Protocol: Step-by-Step Excellence
Consider a standard qPCR workflow where accuracy directly influences scientific validity:
Master Mix Preparation (Critical Step 1)
Create a stock master mix containing:
- DNA polymerase: 0.5 units per reaction
- dNTPs: 200 µM each
- Primers: 200 nM each
- Probes: 100 nM (if using probe-based detection)
- Buffer: appropriate pH and ionic strength
- Total volume: 18 µL per reaction
Equipment selection: Use a P200 electronic micropipette (20–200 µL range) for master mix distribution. Why electronic? The 8-replicate qPCR plate requires 144 µL total master mix (18 µL × 8 reactions). An electronic P200 achieves ±0.8% precision (CV), delivering 144.0±1.15 µL total. A manual P200 achieves ±2.5% precision (CV), delivering 144.0±3.6 µL. The 3x precision improvement translates directly to reduced Ct variance across replicate reactions.
Technique consideration: Use forward pipetting (normal plunger direction) for aqueous master mix. Immerse the tip 3–4 mm into the source tube containing master mix. Depress plunger to first stop, release slowly (1-second aspiration), wait 0.5 seconds for settling, then remove the tip vertically. This standardized technique, when applied consistently by electronic systems, produces coefficient of variation of <1%.

Template Addition (Critical Step 2)
Dispense 2 µL sample DNA template into each well containing 18 µL master mix.
Equipment selection: Use a P20 single-channel electronic micropipette (2–20 µL range). At 2 µL (10% of P20 range), accuracy specifications allow ±3%, but electronic operation reduces actual CV to 0.6%, delivering 2.0±0.015 µL. This precision matters: 2 µL containing 10 ng target DNA gets exactly 10 ng. A 10% variance means some reactions receive 9 ng while others receive 11 ng-not visibly different, yet reflecting a 22% differential in template copy number that compounds through 40 cycles to create detectible Ct differences.
Technique consideration: Reverse pipetting works better for template addition. Depress the P20 plunger fully (past the first stop to the second stop) before immersing the tip. Release to the first stop while immersed in the template source-this aspirates 2 µL plus a small volume overage. Remove the tip. Position the tip at the edge of the receiving well (containing 18 µL master mix) and depress to the first stop only-this dispenses the primary 2 µL volume. The extra aspirated volume remains in the tip, preventing loss to external tip wetting that would reduce actual delivered volume.
Application Impact Metrics
The cumulative effect of precision micropipette work shows clearly in qPCR performance:
- Poorly executed pipetting (manual P1000 for 2 µL additions, CV 5%): Replicate Ct values range ±1.5 cycles
- Adequate technique (manual P20, CV 2%): Replicate Ct values range ±0.7 cycles
- Optimized technique (electronic P20 reverse pipetting, CV 0.6%): Replicate Ct values range ±0.2 cycles
For a study comparing treatment effects expected to produce 1.5 cycle differences, poorly executed pipetting creates data noise that obscures the biological signal. Optimized technique enables clear signal detection with reduced sample size requirements.
Clinical Diagnostics: High-Throughput Standardization
Clinical diagnostics labs process 200–1,000 patient samples daily, demanding speed without sacrificing accuracy. A single misaligned well or improper reagent volume creates false positive/negative results that affect patient care decisions.
ELISA Protocols: The Workhorse Application
Enzyme-linked immunosorbent assay (ELISA) dominates serology and allergen detection across clinical laboratories. A 96-well ELISA plate requires:
- Plate coating (100 µL capture antibody per well, 96 wells = 9,600 µL total)
- Blocking (100 µL per well)
- Sample addition (50 µL per well)
- Primary antibody addition (100 µL per well)
- Secondary antibody addition (100 µL per well)
- Substrate addition (100 µL per well)
- Stop solution addition (50 µL per well)
Operational comparison:
Using single-channel P200 (manual): 45 minutes total time, CV 2.5%, operator fatigue moderate
Using 8-channel P200 (electronic): 6 minutes total time, CV 0.8%, operator fatigue minimal
This 87% time savings across 200 ELISA plates annually (typical diagnostics lab volume) equals 130 labor hours saved-equivalent to 3+ full-time employee weeks. At average laboratory technician salary of $45,000 annually ($22/hour fully loaded cost), this time savings justifies the $6,000–$8,000 equipment investment within a single year purely from labor perspective.
Beyond labor, the precision improvement is consequential. ELISA optical density values (OD) typically range 0.1–2.5 across the dynamic range. An assay with CV 2.5% across replicate wells produces ±0.025–0.063 OD variance. An assay with CV 0.8% produces ±0.008–0.020 OD variance. This 3x reduction in variance improves diagnostic sensitivity-the ability to detect true positives-by enabling lower detection cutoff thresholds.
Cell Culture & Microbiology: Maintenance Precision
Cell culture and microbiology work requires both precision and contamination control. A mammalian cell culture protocol demands media replacement every 48 hours, with volume exchange of 50–80% of the culture vessel volume. While precision matters, preventing cross-contamination becomes equally important.
Sterile Technique in Cell Culture
Culture maintenance requires:
- Media aspiration (remove old media containing metabolic waste)
- Rinsing (add fresh media, then remove-optional, context-dependent)
- Fresh media addition (restore nutrients and maintain osmotic balance)
Equipment selection: Use filter-tip P1000 micropipettes (100–1,000 µL range) with sterile technique:
- Filter tips prevent aerosol contamination of the pipette shaft
- Single-use tips eliminate cross-well contamination risk
- Electronic systems enable consistent slow aspiration (prevents cell resuspension and shearing damage)
Technique specificity: For cell culture media changes, aspiration speed must not exceed 0.5 m/s-faster speeds create shear forces that damage delicate adherent cells. Electronic systems with programmable flow rates enable setting aspiration speed to exactly 0.5 m/s, maintaining consistency across operators and culture vessels. Manual operation risks unintended acceleration if hand speed varies.
Part 4: Best Practices for Optimal Micropipette Performance
Foundational Technique: Forward and Reverse Pipetting
Two primary pipetting techniques address different sample characteristics. Mastering both techniques separates competent laboratory work from optimal results.
Forward Pipetting: Standard Protocol for Aqueous Solutions
Forward pipetting (normal plunger direction) suits most aqueous solutions-buffers, media, reagent solutions. The technique follows this sequence:
- Position the micropipette vertically (90° angle to liquid surface)
- Depress plunger to first stop (pressure equilibration point, where plunger resistance increases perceptibly)
- Immerse tip into source liquid to depth specified by volume:
- 1–10 µL: 2–3 mm immersion
- 10–100 µL: 3–4 mm immersion
- 100 µL: 4–6 mm immersion
- Release plunger slowly (1-second aspiration for small volumes, 2-second for larger volumes)
- Hold position for 0.5–1 second (allows liquid settling in tip)
- Withdraw tip vertically from source liquid
- Position at receiving vessel (can be at angle, typically rim of well)
- Depress plunger to second stop (full plunger depression, goes past first stop)
The sequential timing matters. Releasing the plunger “slowly” means 1 second for small volumes, 2 seconds for large volumes-not instantaneous, yet not so slow that evaporation becomes significant. Waiting 0.5 seconds after aspiration allows the liquid to settle in the tip, eliminating bubbles that would reduce delivered volume.
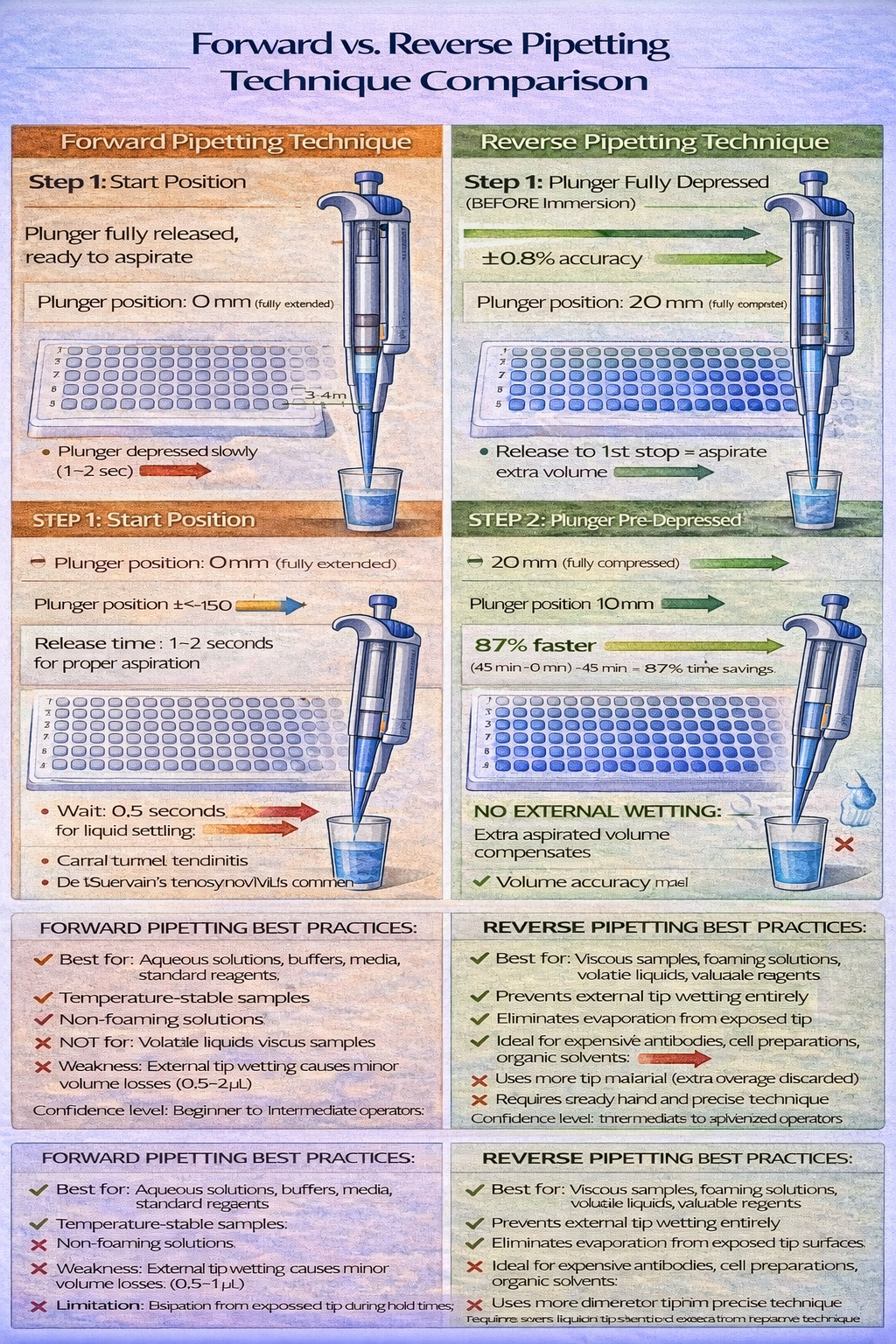
When forward pipetting excels: Aqueous solutions including buffers, media, blood serum, saliva, urine, reagent solutions. Temperature-stable samples. Samples without surface active properties (non-foaming).
When forward pipetting creates problems: Volatile liquids evaporate from the tip between aspiration and dispensing. Foaming reagents trap air bubbles inside the tip. Extremely viscous samples require longer aspiration times. Samples with dissolved gases (media bottled in air) can release gas bubbles during aspiration, creating volume losses.
Reverse Pipetting: Advanced Technique for Difficult Samples
Reverse pipetting (reverse plunger direction) offers advantages for problematic samples. The technique uses plunger positions in opposite sequence:
- Depress plunger fully to second stop (before immersing tip in source)
- Immerse tip into source liquid
- Release plunger only to first stop (aspirate extra volume beyond the target)
- Remove tip from source (extra aspirated liquid remains in tip)
- Position at receiving vessel (insert tip slightly into liquid)
- Depress plunger only to first stop (dispenses target volume only)
- Wipe tip on vessel interior wall (removes external droplet that formed during aspiration)
- Remove tip and discard (the overage volume remains in tip)
This technique prevents external tip wetting that plagues forward pipetting. In standard forward pipetting, liquid coats the external tip surface during aspiration-when transferred to the receiving vessel, gravity causes this liquid to drip into the well (now you’ve delivered 105 µL instead of intended 100 µL). Reverse pipetting completely prevents this error.
When reverse pipetting becomes mandatory: Viscous biological samples (blood sera with high protein content, glycerin solutions, thick cell lysates). Foaming reagents (detergent solutions, culture media). Volatile liquids (DMSO, dimethylformamide). Valuable samples where volume loss is unacceptable (monoclonal antibodies, recombinant proteins, precious research reagents).
Operational disadvantage: Reverse pipetting uses more tip material per pipetting event (you aspirate 105 µL, dispense 100 µL, discard 5 µL) and requires more time. For routine ELISA with 1000+ pipetting events monthly, the cost ($0.02 per extra tip) and time (5+ extra minutes) accumulate. Yet when handling a $2,000/µL monoclonal antibody, the insurance against 5% volume loss justifies the extra time and cost entirely.
Tip Selection & Compatibility Fundamentals
Micropipette tips-the disposable plastic cones that actually contact the liquid-represent both a cost center and a critical performance variable. Tip selection directly impacts accuracy, contamination control, and sample preservation.
Standard Polypropylene Tips: The Laboratory Workhorse
Standard polypropylene tips cost $0.01–$0.03 each, making them economical for high-volume pipetting. They provide universal fit compatibility across most manufacturers’ pipettes (though manufacturers recommend their own tips for optimal performance).
Best use: General laboratory work-buffer preparation, reagent distribution, media handling, non-demanding molecular work.
When standard tips compromise accuracy: Loose fit on tapered cones introduces 1–2% accuracy loss. Some pipette/tip combinations produce unintended air leakage during aspiration. For applications demanding ±1–2% accuracy, manufacturer-specific tips ensure optimal fit.
Low-Retention Tips: Prevention of Precious Sample Loss
Low-retention (hydrophobic) tips use special surface treatment that prevents liquid from adhering to the interior tip surface. When dispensing expensive samples (monoclonal antibodies costing $2,000+/µL, recombinant proteins, precious research compounds), standard tips leave 2–5% of the sample inside the tip. Low-retention tips reduce this to <1%.
The cost multiplier justifies the expense: standard tips cost $0.02, low-retention tips cost $0.15–$0.20. Yet if protecting a $2,000/µL antibody sample, the $0.18 extra cost prevents $20+ in lost sample value. For a research lab using 500 µL of monoclonal antibodies monthly, low-retention tips prevent $100–$200 monthly sample loss-justifying the $10–$15 extra cost per box of tips.
Filter Tips: Contamination Prevention Imperative
Filter tips incorporate an aerosol barrier-a tiny plastic barrier inside the tip that blocks particles from entering the pipette shaft when aerosols form. For PCR work and all RNA/DNA applications, filter tips are non-negotiable. A single contaminated pipette shaft can ruin 100+ experimental reactions through carry-over contamination.
Filter tips cost $0.05–$0.15 each-substantially more than standard tips. Yet the cost is trivial compared to the consequence of contamination. Studies tracking contamination in PCR labs document that 40–60% of non-filter-tip pipettes become contaminated with foreign DNA within 3 months of regular use, creating false positive results in 5–10% of PCR reactions run using those pipettes.
Specialized Tips for Specific Applications
- Wide-bore tips (orifice diameter >1.5 mm): For fragile samples (whole cells, genomic DNA preparations, protein complexes) that shear in standard tips. Wider orifice reduces shear force during aspiration.
- Graduated tips (with volume markings): For visual volume verification in non-automated applications. Reduced accuracy compared to calibrated pipettes (±3–5% typical).
- Extended-length tips (15–20 mm instead of standard 10 mm): For deep well plates or special vessels that require greater tip reach.
Calibration: The Accuracy Verification Foundation
Micropipettes drift out of calibration through three mechanisms: mechanical wear (piston friction, seal degradation), environmental factors (temperature changes, humidity effects), and normal operation (hundreds of thousands of aspiration cycles over years). ISO 8655 standards mandate periodic calibration verification to ensure accuracy remains within specification.
Gravimetric Calibration: The Gold Standard Method
Gravimetric calibration tests actual delivered volumes by weighing dispensed liquid, then converting mass to volume using density factors. This method requires:
Equipment needed:
- Analytical balance (±0.1 mg precision minimum, ±0.01 mg preferred)
- Distilled water at 20±1°C
- Pre-weighed collection containers
- Thermometer (verify temperature for density conversion)
Procedure (10 replicate measurements):
- Calibrate the analytical balance to zero with pre-weighed container
- Pipette 10 replicate volumes of distilled water into the container
- Weigh the container (accurate to 0.1 mg)
- Repeat for 10 separate deliveries into separate pre-weighed containers
- Calculate average mass across replicates
- Convert mass to volume using density conversion:
- At 20°C: 1 mg water ≈ 1.00 µL
- At 25°C: 1 mg water ≈ 1.001 µL
- Compare actual volume to set volume
- Calculate accuracy (% error) and precision (coefficient of variation)
Example calculation:
- Set volume: 100 µL
- 10 replicates weigh: 99.8, 100.1, 99.9, 100.3, 100.0, 99.7, 100.2, 100.1, 99.9, 100.0 mg
- Average: 100.0 mg = 100.0 µL
- Accuracy: (100.0 – 100.0) / 100.0 = 0% error (within spec ±2.5%)
- Standard deviation: 0.18 mg
- Precision (CV): (0.18 / 100.0) × 100 = 0.18% (exceeds spec <2%)
Calibration Frequency Recommendations
- High-use labs (>5 hours pipetting daily): Monthly calibration
- Moderate use (1–5 hours daily): Quarterly calibration
- Low use (<1 hour daily): Semi-annual calibration
- After maintenance/repair: Immediate recalibration before use
- Environmental changes: After significant temperature/humidity shifts (>5°C change, >20% relative humidity change)
Clinical labs and pharmaceutical manufacturers operating under GLP/GMP often maintain calibration records for 7+ years, creating regulatory documentation of instrument performance. For research labs, annual recalibration suffices if usage patterns remain consistent.
Correcting Out-of-Specification Performance
When calibration reveals accuracy outside specification (e.g., consistently delivering 102 µL instead of 100 µL), several corrective actions address the issue:
For mechanical pipettes: Most variable-volume micropipettes include an adjustment screw at the base. Rotating this screw 0.25 turns changes the calibration setpoint by 1–2%. Manufacturer documentation specifies screw direction and rotation increments. Technicians in service-capable labs can self-correct minor drifts. Drifts >2% typically indicate internal seal degradation requiring factory service.
For electronic pipettes: Recalibration is performed through the electronic interface, requiring manufacturer-supplied software or onsite service visits. Most electronic systems incorporate calibration verification functions that compare measured volumes to stored calibration data.
Drift direction interpretation: Consistent under-delivery (pipette delivers <target volume) typically indicates:
- Piston seal degradation (air leakage during aspiration)
- Piston wear (reduced compression efficiency)
- Clogged air vent (creates partial vacuum, reducing aspiration efficiency)
Consistent over-delivery (pipette delivers >target volume) typically indicates:
- Broken or degraded o-ring seal (leakage to higher pressure)
- Mechanical sticking (plunger doesn’t fully release, leaving extra liquid in tip)
- Environmental temperature increase (expanded air volume affects air displacement)

Maintenance & Preventive Care: Extending Equipment Lifespan
Micropipettes engineered and maintained properly function reliably for 10–15 years. Neglected pipettes require replacement within 3–5 years. Preventive maintenance represents a small time investment with substantial payoff.
Daily Maintenance Protocol (5 minutes, post-use)
- External cleaning: Wipe micropipette barrel with lint-free cloth dampened with 70% ethanol
- Tip ejector check: Press ejector button 5–10 times to confirm smooth operation (resistance to pressing indicates jamming)
- Plunger feel verification: Depress and release plunger 5 times to confirm smooth motion without sticking or grinding
- Visual inspection: Check for visible contamination, cracks, or discoloration
- Tip cone inspection: Examine the tip acceptance cone for debris or corrosion
Monthly Maintenance Protocol (15 minutes)
- Internal cleaning (if accessible):
- Remove piston assembly per manufacturer instructions
- Wipe piston and cylinder interior with lint-free cloth dampened with distilled water
- Inspect o-rings for cracks or hardening (indicates aging, consider replacement)
- Reassemble components in reverse order
- Tip cone examination: Inspect under magnification for corrosion, dents, or roughness (visible damage requires factory service)
- Electronic function test (if applicable): Verify button response, display accuracy, battery indicator
- Documentation: Record date and observations in equipment maintenance log
Quarterly/Annual Calibration Cycle (as discussed above)
Part 5: Selection Framework & Procurement Strategy
Comprehensive Decision Matrix: Choosing the Right Micropipette
Selecting appropriate micropipette equipment requires systematic evaluation across multiple criteria. This decision matrix guides selection by application, volume requirements, and operational constraints.
Application × Volume × Throughput Assessment
High-precision, low-throughput applications (research, method development):
- Applications: qPCR setup, enzyme kinetics, DNA quantification, single-sample diagnostics
- Volume: 0.5–100 µL typically
- Throughput: <50 pipetting events daily
- Equipment recommendation: Single-channel electronic P10 or P20 (enables accurate small-volume work) + P200 for larger volumes
- Rationale: Precision matters more than speed; electronic operation enables consistent technique
- Estimated investment: $1,200–$1,800 per set
Moderate-throughput with diverse volumes (mixed research + diagnostics):
- Applications: ELISA batch work, clinical sample prep, culture maintenance
- Volume: 10–1,000 µL across protocols
- Throughput: 100–500 daily pipetting events
- Equipment recommendation: Single-channel set (P20, P200, P1000) + 8-channel electronic P200
- Rationale: Single-channel for precision work, 8-channel for batch operations
- Estimated investment: $4,000–$6,500 per set
High-throughput standardization (clinical diagnostics, screening):
- Applications: 96-well ELISA, high-sample-volume assays, batch testing
- Volume: 20–200 µL primarily
- Throughput: 500–2,000+ daily pipetting events
- Equipment recommendation: Electronic 8/12-channel multichannel system + backup single-channel P200
- Rationale: Multichannel dominates for speed; electronic ensures consistency across operators
- Estimated investment: $8,000–$15,000 per integrated system
Ergonomic Assessment & RSI Prevention
For labs with >2 hours daily manual pipetting, electronic systems become health investments, not luxury upgrades. Calculate 5-year cost of injury:
- Occupational therapy assessment: $1,500
- Carpal tunnel surgery (if required): $10,000–$15,000
- Lost productivity during recovery (6–8 weeks): $8,000–$12,000
- Ergonomic equipment (wrist braces, desk adjustments): $500–$2,000
- Total potential injury cost: $20,000–$39,000
Electronic micropipette system cost: $5,000–$8,000. Even without accounting for time savings, RSI prevention alone justifies the investment.
Budget Allocation Guidelines by Laboratory Type
Academic research labs ($3,000–$6,000 budget):
- Core set: P20 (electronic) + P200 (electronic) + P1000 (manual)
- Rationale: Electronic for precision work where accuracy drives publishable results; manual P1000 sufficient for bulk media/buffer work
- Add 8-channel P200 (mechanical) if processing multiple sample batches
Clinical diagnostics ($8,000–$15,000 budget):
- Core system: Integrated 8-channel electronic + backup P200 electronic
- Rationale: Multichannel for high-volume ELISA/serology work; electronic ensures ISO compliance through consistency
- Add P20 (electronic) for precision work (serum dilution protocols)
Pharmaceutical R&D ($15,000–$30,000+ budget):
- Integrated system: Electronic 8-channel + 96-channel partial-automation + single-channel backup set
- Rationale: Combine manual precision for method development with automated speed for validation experiments
- Add positive displacement system ($2,000–$3,000) for solvent-based assays
Part 6: Advanced Technologies & Future-Focused Solutions
Electronic Micropipette Systems: Features & Advantages
Electronic micropipettes represent 20+ years of innovation aimed at eliminating operator variability. Premium systems now incorporate features that approach fully automated liquid handling.
Key Electronic Features Explained
Volume Lock Function: Prevents accidental volume changes during pipetting. Once set, the electronic display locks the volume. This feature proves invaluable in high-throughput environments where multiple protocols run simultaneously-preventing the common error of grabbing the wrong pipette and pipetting with incorrect volume settings.
Repeat Dispensing Mode: Aspirate once, dispense the same volume multiple times without re-aspiration. For plate filling (50 wells × 50 µL = 2,500 µL aspiration, then 50 separate dispenses), repeat mode eliminates 49 aspiration cycles-reducing execution time from 15 minutes to 3 minutes while improving accuracy since evaporation losses occur only during initial aspiration, not multiple intermediate aspirations.
Customizable Speed Settings: Adjust aspiration/dispensing speed from slow (0.5 m/s, for delicate cells) to fast (5 m/s, for routine aqueous pipetting). This addresses application-specific requirements: cell culture work needs gentle slow aspiration; ELISA plate filling tolerates fast operation. Manual micropipettes can’t adjust speed-operator hand movement creates speed variation.
Data Logging (Premium models): Records every pipetting operation-volume, date, time, pipette ID. For regulated environments (pharmaceutical manufacturing, clinical labs), this creates audit trails demonstrating compliance with documented procedures.
ROI Calculation: When Electronic Systems Pay for Themselves
Scenario: Moderate-throughput clinical diagnostics lab
- Daily ELISA protocols: 10 plates × 96 wells = 960 pipetting operations
- Annual volume: 250,000 pipetting operations
- Manual single-channel setup: 45 min/plate = 7.5 hours daily
- Electronic 8-channel setup: 6 min/plate = 1 hour daily
- Daily time savings: 6.5 hours
- Annual time savings: 1,625 hours (6.5 hours × 250 working days)
- Labor cost savings: $36,563 (1,625 hours × $22.50/hour fully loaded)
- Electronic system investment: $8,000
- ROI timeline: 2.6 months
- 5-year net savings: $176,875 ($36,563 annually × 5 years minus equipment cost)
Beyond pure labor savings, precision improvements reduce rework: electronic systems achieving 0.8% CV vs. manual 2.5% CV eliminate 5–10% of failed assays requiring repeat testing ($500–$1,000 per repeat). This rework elimination typically justifies ROI independently of labor savings.
Automation Integration: When to Pursue Robotic Systems
While individual micropipettes operate at the intersection of precision and manual effort, fully automated liquid handling platforms represent the next evolution. Understanding when automation becomes justified requires analyzing throughput, consistency demands, and available capital.
Semi-Automated Platforms: The Bridge Technology
Semi-automated systems (like Opentrons Open Source liquid handlers or Tecan Freedom EVO) represent an intermediate solution: they automate 60–80% of liquid handling while maintaining flexibility for protocol modifications.
When semi-automation makes sense:
- Throughput: 500–1,000 samples daily (manual becomes overwhelming; full automation not yet justified)
- Protocols: Multiple protocol variations (full automation’s strength is standardized, high-volume single protocols)
- Investment capacity: $50,000–$100,000 available for equipment (less than fully automated systems)
- Technical capability: Staff comfortable with basic programming/troubleshooting
Example application: A pharmacogenomics laboratory processing 700 DNA samples weekly for SNP genotyping could deploy a semi-automated system:
- Manual liquid handler preprocessing: 20% of operations (sample QC, initial dilution)
- Automated platform: 80% of operations (master mix prep, allele-specific PCR setup, plate loading)
- Result: Manual work reduced from 35 hours to 7 hours weekly, but protocol flexibility maintained
Fully Automated Platforms: Industrial-Scale Solutions
Fully automated systems (Tecan Freedom, Hamilton Microlab, Agilent Bravo) manage entire workflows from sample receipt through plate loading for analysis.
When full automation becomes justified:
- Throughput: >2,000 samples daily (automation becomes cost-effective)
- Protocols: Highly standardized, repetitive workflows (strong automation fit)
- Accuracy criticality: Clinical diagnostics, pharmaceutical QA (where consistency is paramount)
- Investment capacity: $150,000–$500,000+ (substantial equipment investment)
ROI calculation example: Pharmaceutical quality assurance lab processing 100 samples daily through a standardized 15-step assay
- Manual approach: 3 technicians × 8 hours × 250 days = 6,000 annual hours @ $50/hour loaded = $300,000 annual labor
- Fully automated system: 1 technician monitoring (0.5 FTE) = $25,000 annual labor
- Annual labor savings: $275,000
- Equipment cost: $250,000
- ROI: 11 months
- 5-year savings: $1,125,000 ($275,000 × 5 years minus equipment cost)
Part 7: Regulatory Compliance & Documentation
ISO 8655 Standards: Understanding Mandatory Specifications
ISO 8655 (Methods for Inspecting and Testing Volumetric Pipetting Devices) establishes performance standards mandatory in regulated laboratory environments. Understanding these standards enables informed procurement and prevents compliance failures.
Performance Criteria Specified by ISO 8655
Accuracy specification by pipette type:
- Class A pipettes (ultra-precise): ±1.5% at nominal volume, ±2% at 50% nominal
- Class B pipettes (standard): ±2.5% at nominal, ±2.5% at 50% nominal
- Class C pipettes (practical use): ±3% at nominal, ±3% at 50% nominal
Most laboratory micropipettes fall into Class B or C. High-precision research applications might specify Class A equipment.
Precision (coefficient of variation) limits:
- Class A: <0.8% CV
- Class B: <1.0% CV
- Class C: <2.0% CV
These specifications appear innocuous until applied to real applications. A clinical lab’s immunoassay requiring ±5% plate-to-plate variance can tolerate Class C equipment. The same lab’s research division developing new assays might require Class A to detect true biological differences within the assay noise floor.
Documentation Requirements for Regulated Environments
Clinical labs operating under CLIA (Clinical Laboratory Improvement Amendments) and CAP (College of American Pathologists) accreditation must maintain:
- Calibration certificates: Maintained for 3–7 years, documenting accuracy verification against standards
- Maintenance logs: Recording cleaning, repairs, component replacements, and any issues
- Personnel training records: Documenting competency verification for each operator
- Equipment specifications: Serial numbers, purchase dates, and usage classifications
- Lot tracking: Serial number assignment to each pipette, enabling contamination tracing if quality issues arise
This documentation burden typically requires 30–60 minutes monthly per pipette set, but proves invaluable when regulatory audits occur or quality investigations require contamination source identification.
Part 8: Troubleshooting Common Issues & Solutions
Even well-maintained micropipettes occasionally encounter problems. Understanding diagnostic approaches accelerates resolution.
| Problem | Observable Indicator | Likely Root Cause | Troubleshooting Steps | Resolution |
|---|---|---|---|---|
| Inconsistent volumes (CV >3%) | Replicates vary by >3% during calibration | Worn piston seal, incorrect tip fit, technique variation | Re-test with different tips; verify technique consistency; recalibrate | Replace o-ring kit ($30–$50); try manufacturer-matched tips; practice technique |
| Liquid dripping after dispensing | 2–5 µL remains hanging from tip exterior after dispensing | External tip wetting, improper tip seal, incorrect dispensing pressure | Use reverse pipetting technique; change tip lot; verify tip compatibility | Practice reverse pipetting; switch to low-retention tips |
| Plunger sticking or grinding | Plunger moves with resistance, creates grinding sound | Contaminant particles in cylinder, piston corrosion, dried residue | Flush cylinder with distilled water; soak piston in distilled water 15 min | Replace piston if corrosion visible; send for professional cleaning |
| Cannot achieve set volume | Micropipette fails to deliver full volume despite plunger depression | Air leakage (worn seal), blocked air vent, broken internal components | Test different tips; inspect air vent opening; verify plunger moves fully | Replace seals if leakage confirmed; clean air vent with pipe cleaner; factory service if structural damage |
| Difficult plunger operation | Requires excessive force to depress plunger | Pressure equalization failure, vent blockage, piston contamination | Check air vent clarity; blow gently through vent opening; clean piston assembly | Clear vent with pipe cleaner; disassemble and clean piston |
| Tips fall off during pipetting | Tip detaches from cone mid-operation | Loose tip fit, bent cone, incompatible tip | Verify tip-cone compatibility; inspect cone for visible bending; try different tip lot | Use manufacturer-matched tips; inspect cone under magnification; factory service if bent |
Strategic Decision Framework
Selecting appropriate micropipette equipment and implementing optimal laboratory practices requires balancing multiple competing factors: accuracy requirements, throughput demands, operator comfort, regulatory obligations, and budget constraints. This guide has provided the technical foundation for informed decision-making.
Key decision frameworks:
- Match micropipette type to sample characteristics: Air displacement for aqueous solutions; positive displacement for organics and viscous samples
- Select volume ranges based on application: Use the smallest micropipette capable of delivering your target volume
- Evaluate electronic vs. manual trade-offs: Electronic systems justify investment through labor savings, precision improvements, and ergonomic benefits in moderate-to-high-throughput labs
- Implement preventive maintenance: Monthly checks and quarterly calibration extend equipment life while maintaining accuracy
- Plan for regulatory compliance: Document all calibrations, training, and maintenance to enable audit readiness
Whether you’re establishing a laboratory from scratch, optimizing existing operations, or upgrading aging equipment, this comprehensive guide provides the technical knowledge and strategic frameworks necessary for excellent decisions that enhance both scientific quality and operational efficiency.
The micropipette you select today will influence research outcomes, diagnostic accuracy, and operator health for years. Choose wisely, maintain diligently, and validate regularly-your laboratory’s results depend on it.